Abstract
Objective: The purpose of this study was to examine the effects of Hormone Replacement Therapy (HRT) on plasma lipoprotein levels, bone mass and the endometrium. Methods: Thirty healthy women were enrolled in the study for climacteric symptoms and received a combination of 17β-estradiol and norethisterone acetate continuously for 2 years. An untreated group, consisting of 25 healthy postmenopausal women presenting the same symptoms and not willing to receive HRT, were also studied and served as controls. Plasma lipoprotein levels, bone mineral density as well as thickness of the endometrium measured by ultrasound and biopsy of the endometrium were evaluated at baseline and after 1 and 2 years. There were no differences between the two groups regarding clinical and demographic baseline data. Results: After 2 years of HRT, total cholesterol and LDL-cholesterol levels were significantly reduced (p<0.05). Changes in serum triglyceride and HDL-cholesterol levels were not statistically significant. Levels of lipoprotein (a) showed a statistically significant decrease at 1st and 2nd year (p<0.05) in both groups while levels of ApoA1 and ApoB were significantly lower only in the HRT group compared to the baseline values. After 2 years of HRT, biomarkers of bone metabolism showed a significant decline, while endometrium was atrophic in 93.3% of the cases. Conclusions: Hormone replacement therapy (17β-estradiol combined with norethisterone acetate) was associated with favorable changes in the lipid status and bone metabolism. Endometrium was atrophic in most of the treated subjects.
The radical drop in estrogen levels met at menopause is associated with an increased risk of osteoporosis and cardiovascular disease. Investigators correlate the incidence of the latter at menopause to that of men1. Epidemiologic and clinical studies have proved this risk to be strongly dependent on lipid and lipoprotein changes2,3. It is also well known that estrogen deficiency increases bone turnover in favor of resorption, resulting in an accelerated rate of bone loss; this loss, accompanied by a subsequent bone microarchitectural deterioration, constitutes a disease status known as osteoporosis4.
Hormone Replacement Therapy (HRT) has a presumed cardioprotective effect due to estrogen beneficial action on lipid profile and vascular endothelium5-7. Estrogens normalize bone metabolism on the skeleton, arrest bone loss and decrease fracture risk4. In order to achieve the above cardioprotective results and prevent osteoporosis, HRT has to be used for an extended period of time, which, however, may be associated with undesired side effects, such as endometrial hyperstimulation resulting in endometrial stromal cells proliferation, and an increased risk of breast cancer8,9.
The aim of this study was to assess the effects of a continuous 2-year combined HRT therapy on the lipid status, bone mineral density and the endometrium.
PATIENTS AND METHODS
This open non-randomized study was carried out in 30 healthy postmenopausal women with climacteric symptoms and 25 untreated. All studied subjects were 1 year menopausal and had serum follicle stimulating hormone (FSH) levels > 30 IU/L.The group of 30 treated women received a continuous combined regimen of replacement therapy consisting of 17™- estradiol 2 mg daily plus norethisterone acetate 1 mg daily (Kliogest, Novo, A/S Denmark) for a period of 2 years.
The untreated group consisted of 25 healthy postmenopausal women, being examined in parallel with the former group but who were not willing to receive HRT. They were suffering the same climacteric symptoms and were not receiving other therapy. This group was designated as a control group.
All patients were evaluated at baseline and at 1 and 2 years. The study protocol was approved by the Ethics Board of our Hospital and all women of the treatment group signed an informed consent form.
Excluded from the study were women with a history of breast malignancy, undiagnosed vaginal bleeding or an endometrial thickness > 5mm (considered as abnormal findings, which needed further investigation); women with thyroid or other endocrine disease; women with hepatic or kidney dysfunction, women taking medication affecting lipid status or bone metabolism; women with a history of thromboembolism; and women taking an estrogen / progestogen regimen during the last 3 months before baseline evaluation. Fasting blood glucose levels were <6.7mmol/l and body mass Index (BMI) was <30. Subjects who discontinued treatment during the 2-year follow-up were also excluded from the study. In fact, although the initial number of participants was 54 in the HRT group, sufficient data were available on only 30 subjects which constituted the study group.
Baseline evaluation included gynecological examination and a PAP smear as well as breast examination and mammography.
Baseline endometrial evaluation consisted of transvaginal sonographic assessment of the endometrium and biopsy with a Novak curette; samples were fixed and evaluated by two pathologists who had no knowledge of the group status of the patient.
Baseline lipid status assessment included determination of total cholesterol (TC), high density (HDL-C) and low density lipoprotein cholesterol (LDL-C), triglycerides (TG), lipoprotein (a) and apolipoproteins A1 and B.
Bone mass measurements: Bone metabolism was evaluated by bone mineral density (BMD) estimation and biochemical markers, such as serum alkaline phosphatase, phosphate, calcium and urinary hydroxyproline/creatinine and calcium/creatinine ratio.
BMD was measured on the lumbar spine (L2 _ L4) using dual energy X_Ray Absorptiometry (DXA Hologic QDR 1000; Hologic Inc.Waltham, MA, USA).
The coefficient of variation in vitro was 0.43%. The coefficient of variation in vivo had been evaluated by comparing 2 measurements performed with the use of a lumbar densitometer at 7-day intervals in 30 volunteers. The coefficient of variation in vivo for the spine was 1.2%.
Laboratory analyses: Fasting blood samples were obtained in EDTA containing tubes between 07.00 and 09.00 hours.
Total cholesterol and triglycerides were assessed enzymatically using an automatic analyzer (Beckman Syncron CX7, Brea, California, USA); HDL-C and LDL-C cholesterol were assessed by electrophoretic methods using the HDL_C supply kit (Helena Laboratory, Beaumont, Texas, USA). Apolipoproteins A1 and B were assessed by adsorption and lipoprotein (a) by enzyme linked immunosorbent assay (Biopool AB, Umea, Sweden).
Plasma glucose was assessed by a glucose oxidase procedure and FSH by an immunofluorometric method.
Statistical analysis:
Data are expressed as mean ± SD. Analysis of variance was used for repeated measurements to compare the concentration of each variable at the end of the 1st and 2nd year of the study, as well as in the untreated group. Statistical significance was inferred at p < 0.05.
RESULTS
There were no statistically significant differences between the two groups regarding the clinical and demographic baseline data (Table 1).
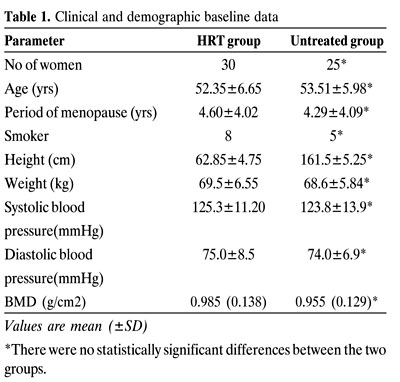
After 2 years of treatment, mean weight gain in the HRT group was 1.6kgr which was not statistically significant different compared to the control group (2.6kgr) p>0.05. Baseline values of systolic and diastolic blood pressure were within normal limits and did not differ significantly between the two groups. After 2 years of follow-up, the blood pressure remained unchanged in the HRT group, while a statistically significant rise was seen in the diastolic blood pressure of the untreated group (p<0.05 – data not shown).
EFFECTS ON LIPID PROFILE
The changes in the lipid profile of the two groups are shown in Table 2.
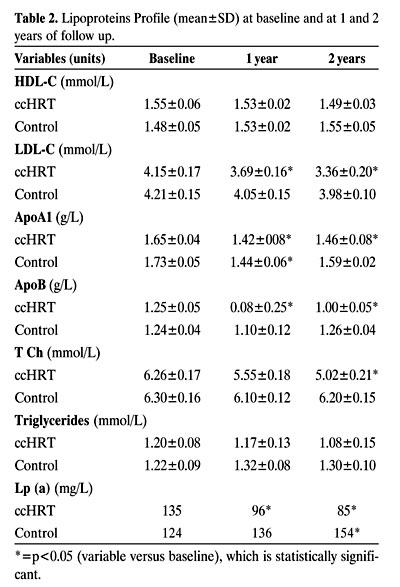
Continuous estrogen_progestogen therapy resulted in significantly reduced levels of serum total and LDL-cholesterol when compared to the corresponding baseline values, while no significant alteration, except for a mild drop in the 2nd year, was observed in serum triglyceride levels.
During the 2-year therapy, HDL-cholesterol levels were mildly but not significantly decreased compared to the corresponding baseline values (p>0.05). Levels of lipoprotein (Lpa) increased significantly in the untreated group over the study period (p<0.05), while in the therapy group, levels showed a statistically significant decrease at the end of the 1st and 2nd year (p<0.05) in comparison to base line values.
Levels of Apo A1 decreased significantly in both groups and this was observed during both the 1st and 2nd year evaluations.
Levels of Apo B were significantly lower compared to the baseline values in the HRT group, while in the control group levels were decreased in the 1st year and then increased during the 2nd year of observation, reaching baseline values.
Effects on the endometrium:
Six women of the study group experienced vaginal bleeding during the first 3 months of therapy, 3 women between the 4th and 6th and 2 between the 7th and 9th month. None of the women in the untreated group experienced bleeding.
Endometrial biopsy:
At the end of treatment, atrophic endometrium was the most common finding in both groups, being observed in 93.33% (n=28) and 92% (n=23) of the HRT and untreated subjects, respectively. Curretings were not performed in 2 patients from each group.
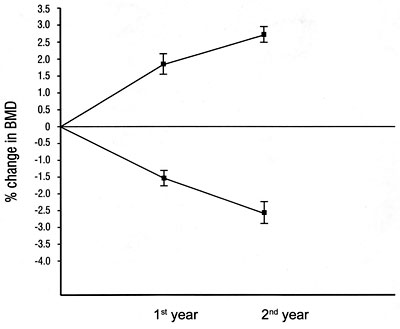
Figure 1.Mean percentage change in spine bone mineral density (BMD) in the 2 study groups during the 1st and the 2nd year.
Effects on spine BMD: At the beginning of the study, there was no significant difference concerning BMD between the two groups (Table 1). In the HRT group, there was a statistically significant increase by 2.8% in the lumbar BMD (Figure 1) in comparison with baseline values (p<0.05). On the contrary, a significant decrease by 2.6% (p<0.05) after 1 and 2 years was observed in the untreated group, compared to baseline values. Differences in BMD values between the two groups were statistically significant (p<0.05).
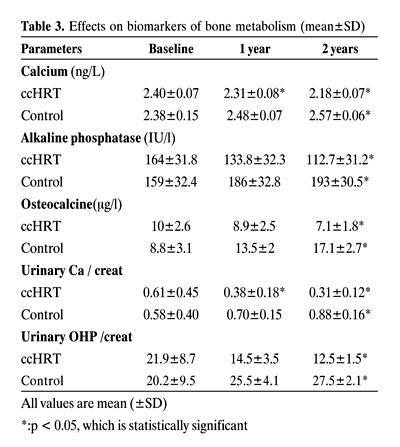
Analysis of biomarkers of bone metabolism showed statistically significant declines in serum phosphate, alkaline phosphatase, serum calcium and urinary hydroxyproline/creatinine and calcium/creatinine ratio in the HRT group (Table 3). Conversely, a significant increase was seen on bone biomarkers of bone metabolism in the control group.
DISCUSSION
Coronary heart disease (CHD) is a major cause of death in postmenopausal women. Epidemiologic studies have established lipid and lipoprotein aberration as risk factors for the CHD. Plasma total cholesterol is directly related to mortality from CHD10, while elevated plasma triglyceride levels have also been considered as an independent risk factor11. It has been shown that a 1% increase in total cholesterol or LDL increases the risk of CHD by 2%, while a 1% reduction in HDL increases this risk by 2-4.7%. Furthermore, a 1% decrease in LDL-C corresponds to a 2% decrease in CHD12. The approximately 20% decrease in LDL-C, observed in our results, suggests a probable 40% decrease in the risk of CHD.
In our study, HRT therapy resulted in a favorable lipid status as seen by the significant reduction in total and LDL-cholesterol and a mild reduction in the triglyceride levels, although HDL was mildly decreased. These results agree with those reported in other studies using similar combination of continuous combined HRT13-15. However, other investigations have reported no changes in the triglyceride levels13.
Apolipoprotein A1, a major component of HDL cholesterol, is inversely related to cardiovascular disease16, whereas Apolipoprotein B is associated with LDL and is directly correlated with CHD17. Lipoprotein(a) has been shown to act as an independent risk factor for premature atherosclerosis and CHD18. In our study, HRT resulted in a drop in apolipoprotein B and lipoprotein (a) and, although these changes appear favorable, HDL and apolipoprotein A1 reductions seem potentially harmful regarding CHD. These changes are probably due to the androgenic effect of norethisterone.
Most of the studies reported in the literature assess changes in lipid metabolism over a 6 to 12_month period of HRT treatment19. We consider that the relatively prolonged duration of treatment in our group, as well as the inclusion of a parallel-untreated group, potentiates the validity of our results.
No significant gain in body weight was found in our study, which is in accordance with the findings of other investigators20. Furthermore, the increase in the diastolic blood pressure occurring in postmenopausal women was not detected in our HRT group, a finding that may be attributed to the replacement therapy since it was observed in the untreated group. Reports on non-lipid determinants of CHD risk with continuous combined HRT indicate a potentially favorable reduction in plasma endothelin-121,22 and a neutral effect on haemostasis23,24 with HRT.
It is well known that postmenopausally there is a loss of bone mass at a rate of 1 to 3% annually for the first 5 years following the last menstrual period. Continuous combined HRT has an anti-resorptive effect and, as a result, bone loss is arrested with a consequent decrease in fracture risk in all major skeletal sites. In our study, a statistically significant increase in the lumbar BMD was noted, a result that was corroborated by the biomarker analysis. These findings are in agreement with those reported by other authors using HRT in postmenopausal women13.
Undoubtedly, further investigation is needed, especially with regard to the necessary period of therapy and side effects on the breast since recent studies make no mention of difference between HRT and placebo in the outcomes for secondary prevention of heart disease25 nor of the still existing risks of breast cancer associated with these regimens26.
REFERENCES
1. Ross RK, Paganini-Hill A, Mack TH, et al, 1989 Cardiovascular benefits of estrogen therapy. Am J Obstet Gynecol 160:1301-1306.
2. Martin MJ, Hulley SB, Browner WS, et al, 1986 Serum cholesterol, blood pressure and mortality: implications from a cohort of 361,661 men. Lancet 2: 933-936.
3. Stamler J, Wentworth D, Neaton JD, 1986 Is the relationship between serum cholesterol and risk of premature death from coronary heart disease continuous and graded?. JAMA 256: 2823-2828.
4. Lindsay R, Hart DM, Forrest C, Baird C, 1980 Prevention of spinal osteoporosis in oophorectomised women. Lancet 1: 1151-1153.
5. Bush TL, Barrett-Connor E, Cowan LD, et al, 1987 Cardiovascular mortality and noncontraceptive use of estrogen in women. Results from the lipid research clinics program follow-up study. Circulation 75: 1102-1109.
6. Sullivan JM, Zwaag RV, Lemp GF, et al, 1988 Postmenopausal estrogen use and coronary atherosclerosis. Ann Intern Med 108: 358-363.
7. Witteman JCM, Grobbee DE, Kok FJ, et al, 1989 Increased risk of atherosclerosis in women after menopause. Br Med J 298: 642-644.
8. Smith DC, Prentice R, Thompson DJ, et al, 1975 Association of exogenous estrogen and endometrial carcinoma. N Engl J Med 293: 1164-1167.
9. Whitehead MI, King RJB, McQueen J, et al, 1979 Endometrial histology and biochemistry in climacteric women during estrogen and estrogen/progestogen therapy. J R Soc Med 72: 322-327.
10. Qizibash N, Jones L, Warlow C, Mann J, 1991 Fibrinogen and lipid concentration as risk factors for transient ischaemic attacks and minor ischaemic strokes. BMJ 303: 605-609.
11. Austin M, 1989 Plasma triglycerides as a risk factor for coronary heart disease. The epidemiological evidence and beyond. Am J Epidemiol 129: 149-159.
12. Lipid Research Clinics Program, 1984 The Lipid Research Clinics coronary, primary prevention trial results. The relationship of reduction in incidence of coronary heart disease to cholesterol lowering. JAMA 251: 365-374.
13. Christiansen C, Riis BJ, 1990 Five years with continuous combined oestrogen/progestogen therapy. Effects on calcium metabolism, lipoproteins and bleeding pattern. BJOG 97: 1087-1092.
14. Sporrong T, Hellgren M, Samsioe G, Mattsson LA, 1989 Metabolic effects of continuous estradiol-progestin therapy in postmenopausal women. Obstet Gynecol 73: 754-758.
15. Milner MH, Sinnott MM, Cooke TM, Kelly A, McGill T, Harrison RF, 1996 A 2-year study of lipid and lipoprotein changes in postmenopausal women with tibolone and estrogen-progestin. Obstet Gynecol 87: 593-599.
16. Contois JH, McNamara JR, Lammi-Keefe CJ, Wilson PW, Massov T, Schaefer EJ, 1996 Reference intervals for plasma apolipoprotein B determined with a standardized commercial adsorption assay. Results from the Framingham Offspring Study. Clin Chem 42: 507-514.
17. Contois JH, McNamara JR, Lammi-Keefe CJ, Wilson PW, Massov T, Shaefer EJ,1996 Reference intervals for plasma apolipoprotein B determined with a standardilised commercial adsorption assay. Results from the Framingham Offspring Study. Clin Chem 42: 515-523.
18. Scanu AM, 1992 Lipoprotein (a): a genetic risk factor for premature coronary heart disease. JAMA 267: 3326-3329.
19. Creatsas G, Arefetz N, Adamopoulos PN, Konstantellou E, Aravantinos D, 1994 Transdermal estradiol plus oral medroxyprogesterone acetate replacement therapy in primary amenorrheic adolescents. Clinical, hormonal and metabolic aspects. Maturitas 18: 105-114.
20. Hassager C, Christiansen C, 1988 Blood pressure during oestrogen / progestogen substitution therapy in healthy postmenopausal women. Maturitas 9: 315-323.
21. Ylicorkala O, Orpana A, Puolakka J, Pyorata T, Viinikka L, 1995 Postmenopausal hormonal replacement decreases plasma levels of endothelin-1. J Clin Endocrinol Metab 80: 3384-3386.
22. Creatsas G, Malamitsi-Puchner A, Hassan E, Economou E, Aravantinos D, 1996 Endothelin plasma levels in primary amenorrheic adolescents before and after estrogen treatment. J Soc Gynecol Invest 3: 350-353.
23. Sporrong T, Mattsson LA, Samsie G, Stigendal L, Hellgren M, 1990 Haemostatic changes during continuous estradiol-progestogen treatment of postmenopausal women. BJOG 97: 939-944.
24. Habiba M, Akkas A, AL-Azzawi F, 1995 Effect of a new cyclical sequential postmenopausal HRT on lipoprotein, apoprotein and thrombophilia profile . Eur J Obstet Gynecol Reprod Biol 62: 89-94.
25. Hulley S, Grady D, Bush T, et al, 1998 Randomized trial of estrogen plus progestin for secondary prevention of coronary heart disease in postmenopausal women. Heart and Estrogen/progestin Replacement Study (HERS) Research Group. JAMA 280: 605-613.
26. Speroff L, 2000 Postmenopausal estrogen-progestin therapy and breast cancer: a clinical response to epidemiologic reports. Climacteric 3: 3-12.
Address correspondence and requests for reprints to:
Charalambos Siristatidis, 17 Didimotichou St., ATHENS 13671, GREECE,
Tel. +03/01/2432664, Fax +03/01/7233330, e-mail: harrysiri@hotmail.com
Received 19-04-02, Revised 26-06-02, Accepted 25-08-02