Abstract
Objective: Coronary artery disease (CAD) risk increases in women after the menopause. The aim of this study was to determine the effect of diabetes on the severity of CAD in postmenopausal women undergoing coronary angiography. Design: 180 postmenopausal women underwent coronary angiography for suspected CAD. CAD severity was assessed by the number of arteries (0-3) with >50% stenosis in the angiography. Forty-four women had type 2 diabetes mellitus (T2DM). Predisposing risk factors and biochemical and hormonal parameters were recorded. The diabetic women were older (p=0.014), had higher BMI and waist circumference (p<0.001), higher prevalence of hypertension (p=0.002), higher levels of triglycerides, uric acid and higher HOMA-Insulin Resistance Index (p=0.009). Results: The women with diabetes had a higher prevalence of severe stenosis in the angiography: T2DM: 0-vessels 25%, 1-vessel disease 18.2%, 2-vessels disease 22.7%, 3-vessels disease 34.1%, vs. 49.2%, 23.5%, 22.1%, 5.1% in the non-diabetic women, respectively (p<0.001). Binary logistic regression analysis showed that T2DM was a significant predictor of severe CAD (≥3 vessel disease) independently of age, family history of T2DM, BMI, time since menopause, hypertension and hypercholesterolemia. Women with T2DM also had lower sex hormone binding globulin (SHBG, p=0.010) levels compared to non-diabetic women. Conclusions: Diabetic postmenopausal women develop more severe CAD compared to non-diabetic women. This association is independent of other predisposing factors and suggests an independent effect of T2DM on the atherosclerotic process, at least in women after menopause.
INTRODUCTION
Coronary artery disease (CAD) is the main cause of death in women and this risk increases after the menopause.1-4 Premenopausal women have less severe coronary artery stenosis in the angiography than postmenopausal women5 and are at lower risk for CAD than men.1 This disparity suggests a protective effect of endogenous estrogens against atherosclerosis, whereas their deprivation aggravates the atherosclerotic process after the menopause.4
The rising incidence of CAD during menopause occurs in parallel with an increase in the incidence of both type 2 diabetes mellitus (T2DM) and hypertension. The transition from premenopausal to postmenopausal status is associated with the emergence of various risk factors for metabolic syndrome as well as CAD, including increased central adiposity, a more atherogenic lipid profile and increased glucose and insulin levels.4 The presence of diabetes increases the risk for CAD in both premenopausal and postmenopausal women6 and probably counteracts the protective effect of estrogen on the vasculature6,7 so that premenopausal diabetic women show the same risk for CAD as men.8
On the other hand, CAD is considered as one of the most important complications of diabetes mellitus (DM) in all types of DM and in both sexes. Hypertension and dyslipidemia are risk factors for CAD among diabetic patients and it is well established that patients with diabetes have more extensive and more rapidly progressive CAD than non-diabetic subjects.9-13 The Framingham study as well as other studies14-16 showed a 2 to 4-fold higher prevalence of atherosclerotic disease in diabetic compared to non-diabetic individuals. Diabetic women have greater mortality risk from coronary heart disease than non-diabetic men and women.17
The aim of our study was to evaluate the effect of diabetes on the severity of CAD, as well as its relation with other metabolic and hormonal factors, in postmenopausal women undergoing coronary angiography.
SUBJECTS AND METHODOLOGY
We included 198 consecutive postmenopausal women (age 45-88 yrs) who were referred over a period of approximately three years (from January 2004 to October 2006) to our institution for elective coronary angiography, performed for the following reasons: angina, atypical chest pain, positive stress test, previous acute myocardial infarction, dyspnoea during exercise, strong family history for coronary heart disease and valve disease. Exclusion criteria were: previous coronary artery bypass surgery or angioplasty, type 1 DM, organ transplantation, renal haemodialysis and long-term use of corticosteroids, hormone replacement therapy (HRT), use of immunosuppressive or chemotherapeutic drugs as well as Cushing’s disease and acromegaly. One woman had undergone heart transplantation, one had acromegaly, four women were on long-term therapy with corticosteroids or immunosuppressive drugs, seven were premenopausal and a further five women refused to participate in the study. Finally, 180 postmenopausal women were included in the study. Five of them underwent coronary angiography a few days after an acute coronary attack. The study was approved by the institutional Ethics Committee and all subjects gave their informed consent.
The severity of CAD was assessed by the number of arteries (0, 1, 2 or 3) with more than 50% reduction of the luminal diameter of the coronary arteries in the angiography. The number of affected vessels (0, 1, 2 or 3) was recorded and confirmed by two independent cardiologists.3
Clinical history was recorded, including chronological age and age at onset of CAD as well as clinical parameters of cardiovascular disease: presence of angina diagnosed according to clinical symptoms and electrocardiographic findings, past myocardial infarctions, presence of family history for premature CAD (<55 years), diabetes mellitus and other risk factors for CAD such us hypertension, dyslipidemia and smoking. Current drug therapy was also recorded.
Type 2 diabetes mellitus (T2DM) was defined as fasting blood glucose levels greater than 126 mg/dl (7 mmol/L) and impaired fasting glucose as fasting blood glucose levels 100-125 mg/dl (5.6-6.9 mmol/L) on two occasions during the current visit or if previously diagnosed according to the American Diabetes Association criteria.18 The presence of hypertension was defined as systolic and/or diastolic blood pressure higher than 139 mmHg and/or 89 mmHg, respectively, for non-diabetic and higher than 129/79 mmHg for diabetic patients.19
Cigarette smoking was defined as current and recently ceased smoking, that is, having ceased less than 6 months previously. Hyperlipidemia was defined as total blood cholesterol of more than 200 mg/dl (5.17 mmol/L) or low density cholesterol (LDL) of more than 160 mg/dl (4.14 mmol/L) in patients with less than two cardiovascular risk factors, or more than 130 mg/dl (3.36 mmol/L), in patients with two or more cardiovascular risk factors, or more than 100 mg/dl in patients with CAD or diabetes. Hypertriglyceridemia was defined as total blood triglycerides of more than 150 mg/dl (2.30 mmol/L).20 To avoid colinearity problems, a CAD-risk score was calculated for each patient including all the factors which might be correlated, such as hypertension, hyperlipidemia and smoking. This calculated risk was then considered as a possible confounding factor in the association between T2DM and the severity of CAD.
A detailed gynaecological and reproductive history concerning age at menarche and menopause, number of menstrual cycles per year, use of hormone replacement therapy (HRT) and/or contraceptive medication were also recorded for each patient.
At clinical examination height and weight were measured with women wearing indoor clothes without shoes. Body mass index (BMI), calculated as weight (kg)/height (m2), was used as a measure of obesity and waist perimeter and waist to hip ratio (cm) were used to evaluate fat distribution. Systolic and diastolic blood pressure was calculated as the mean of two measurements using a random zero sphygmomanometer with women in the sitting position.
The clinical characteristics as well as the presence of various risk factors for CAD in the study population were as follows: Mean age 65.91±0.7 years (range 45-88yrs), smoking 24.6%, positive family history for CAD at a young age (<55 years) 45.3%, and a positive family history for diabetes 24.5%. Among the patients, 25.6% reported a history of previous myocardial infarction, 73.5% a history of angina, 35.6% hypertriglyceridemia, 76.9% hypercholesterolemia and another 66.3% a history of hypertension. The mean BMI was 28.2±0.3 and the waist to hip ratio (WHR) 0.9±0.006. Among diabetic subjects 4.5% were being treated with insulin and 16.3% with glucose lowering pills.
Fasting blood samples were obtained by venipuncture between 08:00-09:00h. Biochemical parameters (glucose, cholesterol, HDL, LDL, triglycerides, uric acid) were measured immediately using an automated analyser Integra 400 (Roche). Specimens were kept frozen at -20o until analysis of 17β-estradiol, SHBG, testosterone and insulin levels. 17β-estradiol was measured using the Spectria E2 sensitive method [(Orion Diagnostics, Finland), (inter-assay CV=5.2% and intra-assay CV=4.2%)], sex hormone binding globulin (SHBG) was determined by IRMA (Radim S.p.A, Via del Mare, Domecia Roma, Italia), inter-assay CV=8.1% and intra-assay CV=5%. Serum insulin was determined by IRMA [(Biosource Europe SA, Nivelles, Belgium) (intra- and inter-assay CV 7.8% and 6.4%, respectively, at 100 μU/ml)]. Total serum testosterone was measured by RIA (Biosource Europa SA, Nivelles, Belgium), with a reference range of 0.35 – 2.8 nmol/L. Interassay CV=6.2% and intra-assay CV=4.6%.
Basal insulin resistance index (Homeostasis Model Assessment, HOMA) was calculated according to the formula: Insulin resistance = FI X G/22.5, where FI=fasting insulin (μU/ml) and G=fasting glucose (mmol/L).21 Patients who were treated with insulin were excluded from the final analysis (n=2).
STATISTICAL ANALYSIS
All descriptive data are expressed as mean ± SE. Statistical analysis was carried out using the SPSS statistical package (version 11.5). The parameter showing the number of diseased vessels was transformed into a binary parameter as follows: “three vessel disease” Yes or No according to the presence of ≥3 vessels or <3 vessels with severe stenosis. Multivariate binary logistic regression analysis was used to identify the predictors of patients with 3 vessel disease. The multivariate model included all variables associated with the binary parameter with a p value of less than 0.2 by univariate logistic regression analysis.
The chi-square test with Yates’ continuity correction was used, or chi-square for linear association as appropriate. Student’s t-test was used to compare mean values between groups where the distribution was normal.
RESULTS
According to the results of the angiography, 78 women had 0 vessel disease, 40 had 1, 40 had 2 and 22 had 3 vessel disease. Forty-four of these women (24.4%) had pre-existing type 2 diabetes mellitus, while the remaining 136 women did not have diabetes and served as the control group.
The women with diabetes had a higher prevalence of severe stenosis in the angiography compared with the non-diabetic women (Figure 1). Among the diabetic women, 25% had 0 affected vessels, 18.2% had 1 vessel, 22.7% had 2 and 34.1% had 3 vessels with >50% stenosis; the corresponding prevalence in the non-diabetic women were 49.2%, 23.5%, 22.1%, and 5.1% (p <0.001, Pearson’s chi-square). When the non-diabetic women were separated into two groups according to the fasting glucose levels, the relevant prevalence for women with fasting glucose levels less than 100 mg/dl (5.5 mmol/L) were 54.7%, 25.6%, 15.1% and 4.6%, respectively. The non-diabetic women with fasting glucose between 100 and 125 mg/dl (5.5 – 6.9 mmol/L) also had a lower prevalence of severe stenosis in the angiography compared with the diabetic women (x2=9.8, p=0.019), but there was no significant difference in the severity of CAD between this group and the group of non-diabetic women with fasting glucose levels less than 100 mg/dl [5.5 mmol/L (x2=7.18, p=0.066, Tables 1 , 2 )].
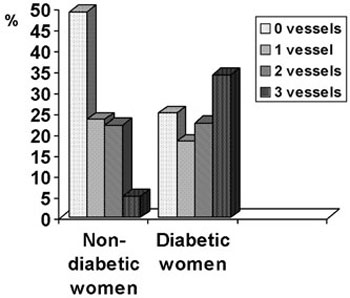
Figure 1. Distribution (%) of vessels with severe stenosis in the angiography according to the presence or absence of diabetes in postmenopausal women undergoing coronary angiography (Pearson’s chi-square test, p <0.001).
The diabetic women were significantly older, had higher BMI, higher waist circumference and WHR. They had more frequently a family history of T2DM, higher prevalence of hypertension and of hypertriglyceridemia as well as higher levels of triglycerides and of uric acid. HOMA-Insulin Resistance Index as well as insulin levels were higher in the group of diabetic women (Table 3). There were no statistically significant differences in smoking, use of lipid lowering drugs, family history for CAD, the age of appearance of CAD, nor in systolic and diastolic arterial blood pressure between the two groups. Diabetic subjects had lower high density lipoprotein (HDL) levels, the difference however did not reach statistical significance. There was no statistically significant difference in the incidence of myocardial infarction or angina between the two groups (Table 3 ). By binary logistic regression analysis, patients with T2DM were at higher risk of having 3- vessel CAD as compared with patients without T2DM. This association was independent of other well established risk factors (Table 4 ).
Finally, there were no statistically significant differences between the two groups as to the age of menopause and the time since menopause. There was no statistically significant correlation between diabetic status and sex hormone levels (estrogen and testosterone). Women with diabetes had lower SHBG levels than non-diabetic women (p=0.012).
DISCUSSION
This is the first study which examines the influence of diabetes on the severity of coronary artery disease in a highly selected group of postmenopausal women undergoing coronary angiography with concomitant investigation of reproductive and hormonal para¬meters. We found that diabetic women have more severe CAD, expressed as the number of affected vessels with greater than 50% stenosis. This association was independent of other risk factors for CAD such as chronological age, family history of T2DM, smoking, hypertension and hyperlipidemia, as well as time since menopause.
Our findings are analogous to those of other reports in the literature concerning the influence of diabetic status on CAD severity22-26 and on the progression of CAD in the angiography9,27-29 in diabetic patients of both sexes. A very recent study found that the presence of diabetes was a predictor of the progression of CAD in the angiography in postmenopausal women.30 Furthermore, other studies which investigated large population samples of women showed that the severity of CAD assessed by coronary angiography was associated with diabetes independently of other predisposing factors,31,32 while another study showed that diabetic women <55 years of age had CAD findings comparable to those of older non-diabetic women.33 However, such findings are not unanimous, as there have been a few studies in the literature that have found no associations between the presence of diabetes and the severity of CAD in patients with cardiovascular disease.34 Similarly to other studies, we did not find a significant difference in the severity of CAD between non-diabetic women with fasting glucose levels between 100 and 125 mg/dl (5.5 – 6.9 mmol/L) and those with less than 100 mg/dl (5.5 mmol/L).26,35
We also found that other risk factors, such us hypertension, obesity, indices of central adiposity and insulin resistance as well as hypertriglyceridemia, were more frequent in the group of diabetic patients. We did not find any additive effect of age at menopause or time since menopause on the severity of CAD in women with T2DM. As far as we know, there is no evidence that menopause affects the appearance of T2DM, although there are reports in the literature showing a possible association of insulin resistance with menopausal status;36 this finding, however, is not consistent.37
So far as the basal mechanism of the atherosclerotic process is concerned, this appears to be roughly similar in diabetic and non-diabetic populations. Specifically, in diabetic subjects, hyperinsulinemia, hyperglycemia, oxidative stress and abnormal protein glycosylation possibly aggravate the atherosclerotic process.6,38
Another finding of our study was the lower levels of SHBG in the diabetic group, a finding that has also been reported in previous studies.39 Especially in women, the increased CAD risk,40,41 the risk of developing diabetes42 as well as various predisposing risk factors for CAD have been strongly associated with lower SHBG levels.43,44 This may be due to the association of SHBG with insulin resistance and the metabolic syndrome in women.45,46 It should be remembered that lower SHBG levels also serve as an index of androgenicity in women as they affect the levels of free androgens, which are considered to be detrimental for the vasculature in women.46,47
In conclusion, our results show that diabetic postmenopausal women have more severe coronary artery disease compared to non-diabetic women when evaluated by coronary angiography. This association is stronger than most of the other classical predisposing factors and suggests an independent effect of T2DM on the atherosclerotic process, at least in women after the menopause. The suggested multi-targeted treatment of all risk factors for CAD48 in diabetic patients appears to be even more justified in postmenopausal women.
REFERENCES
1. Isles CG, Hole DJ, Hawthorne VM, Lever AF, 1992 Relation between coronary risk and coronary mortality in women of the Renfrew and Paisley survey: comparison with men. Lancet 339: 702-706.
2. Colditz GA, Willett WC, Stampfer MJ, Rosner B, Speizer FE, Hennekens CH, 1987 Menopause and the risk of coronary heart disease in women. N Engl J Med 316: 1105-1110.
3. Saltiki K, Doukas C, Kanakakis J, Anastasiou E, Mantzou E, Alevizaki M, 2006 Severity of cardiovascular disease in women: relation with exposure to endogenous estrogen. Maturitas 55: 51-57.
4. Saltiki K, Alevizaki M, 2007 Coronary heart disease in postmenopausal women; the role of endogenous estrogens and their receptors. Hormones (Athens) 6: 9-24.
5. Gurevitz O, Jonas M, Boyko V, Rabinowitz B, Reicher-Reiss H, 2000 Clinical profile and long-term prognosis of women < or = 50 years of age referred for coronary angiography for evaluation of chest pain. Am J Cardiol 85: 806-809.
6. Sowers JR, 1998 Diabetes mellitus and cardiovascular disease in women. Arch Intern Med 158: 617-621.
7. Kaseta JR, Skafar DF, Ram JL, Jacober SJ, Sowers JR, 1999 Cardiovascular disease in the diabetic woman. J Clin Endocrinol Metab 84: 1835-1838.
8. Steinberg HO, Paradisi G, Cronin J, et al, 2000 Type II diabetes abrogates sex differences in endothelial function in premenopausal women. Circulation 101: 2040-2046.
9. Alderman EL, Corley SD, Fisher LD, et al, 1993 Five-year angiographic follow-up of factors associated with progression of coronary artery disease in the Coronary Artery Surgery Study (CASS). CASS Participating Investigators and Staff. J Am Coll Cardiol 22: 1141-1154.
10. Pan WH, Cedres LB, Liu K, et al, 1986 Relationship of clinical diabetes and asymptomatic hyperglycemia to risk of coronary heart disease mortality in men and women. Am J Epidemiol 123: 504-516.
11. Kanaya AM, Grady D, Barrett-Connor E, 2002 Explaining the sex difference in coronary heart disease mortality among patients with type 2 diabetes mellitus: a meta-analysis. Arch Intern Med 162: 1737-1745.
12. Folsom AR, Szklo M, Stevens J, Liao F, Smith R, Eckfeldt JH, 1997 A prospective study of coronary heart disease in relation to fasting insulin, glucose, and diabetes. The Atherosclerosis Risk in Communities (ARIC) Study. Diabetes Care 20: 935-942.
13. Miettinen H, Lehto S, Salomaa V, et al, 1998 Impact of diabetes on mortality after the first myocardial infarction. The FINMONICA Myocardial Infarction Register Study Group. Diabetes Care 21: 69-75.
14. Kannel WB, McGee DL, 1979 Diabetes and cardiovascular disease. The Framingham study. JAMA 241: 2035-2038.
15. Haffner SM, Lehto S, Ronnemaa T, Pyorala K, Laakso M, 1998 Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 339: 229-234.
16. Eckel RH, Kahn R, Robertson RM, Rizza RA, 2006 Preventing cardiovascular disease and diabetes: a call to action from the American Diabetes Association and the American Heart Association. Diabetes Care 29: 1697-1699.
17. Barrett-Connor EL, Cohn BA, Wingard DL, Edelstein SL, 1991 Why is diabetes mellitus a stronger risk factor for fatal ischemic heart disease in women than in men? The Rancho Bernardo Study. JAMA 265: 627-631.
18. Genuth S, Alberti KG, Bennett P, et al, 2003 Expert Committee on the Diagnosis and Classification of Diabetes Mellitus. Follow-up report on the diagnosis of diabetes mellitus. Diabetes Care 26: 3160-3167.
19. Chobanian AV, Bakris GL, Black HR, et al, 2003 Seventh report of the Joint National Committee on Prevention, Detection, Evaluation, and Treatment of High Blood Pressure. Hypertension 42: 1206-1252.
20. Executive Summary of The Third Report of The National Cholesterol Education Program (NCEP) Expert Panel on Detection, Evaluation, And Treatment of High Blood Cholesterol In Adults (Adult Treatment Panel III), 2001 JAMA 285: 2486-2497.
21. Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC, 1985 Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28: 412-419.
22. Henry P, Makowski S, Richard P, et al, 1997 Increased incidence of moderate stenosis among patients with diabetes: substrate for myocardial infarction? Am Heart J 134: 1037-1043.
23. Freedman DS, Gruchow HW, Bamrah VS, Anderson AJ, Barboriak JJ, 1988 Diabetes mellitus and arteriographically-documented coronary artery disease. J Clin Epidemiol 41: 659-668.
24. Lemp GF, Vander Zwaag R, et al, 1987 Association between the severity of diabetes mellitus and coronary arterial atherosclerosis. Am J Cardiol 60: 1015-1019.
25. Fallow GD, Singh J, 2004 The prevalence, type and severity of cardiovascular disease in diabetic and non-diabetic patients: a matched-paired retrospective analysis using coronary angiography as the diagnostic tool. Mol Cell Biochem 261: 263-269.
26. Ledru F, Ducimetiere P, Battaglia S, et al, 2001 New diagnostic criteria for diabetes and coronary artery disease: insights from an angiographic study. J Am Coll Cardiol 37: 1543-1550.
27. Vanhaecke J, Piessens J, Van de Werf F, Willems JL, De Geest H, 1983 Angiographic evolution of coronary atherosclerosis in non-operated patients. Eur Heart J 4: 547-556.
28. Raichlen JS, Healy B, Achuff SC, Pearson TA, 1986 Importance of risk factors in the angiographic progression of coronary artery disease. Am J Cardiol 57: 66-70.
29. Manson JE, Colditz GA, Stampfer MJ, et al, 1991 A prospective study of maturity-onset diabetes mellitus and risk of coronary heart disease and stroke in women. Arch Intern Med 151: 1141-1147.
30. Mellen PB, Cefalu WT, Herrington DM, 2006 Diabetes, the metabolic syndrome, and angiographic progression of coronary arterial disease in postmenopausal women. Arterioscler Thromb Vasc Biol 26: 189-193.
31. Sharaf BL, Pepine CJ, Kerensky RA, et al, 2001 Detailed angiographic analysis of women with suspected ischemic chest pain (pilot phase data from the NHLBI-sponsored Women’s Ischemia Syndrome Evaluation [WISE] Study Angiographic Core Laboratory). Am J Cardiol 87: 937-941.
32. Natali A, Vichi S, Landi P, Severi S, L’Abbate A, Ferrannini E, 2000 Coronary atherosclerosis in Type II diabetes: angiographic findings and clinical outcome. Diabetologia 43: 632-641.
33. Melidonis A, Dimopoulos V, Lempidakis E, et al, 1999 Angiographic study of coronary artery disease in diabetic patients in comparison with nondiabetic patients. Angiology 50: 997-1006.
34. Pajunen P, Nieminen MS, Taskinen MR, Syvanne M, 1997 Quantitative comparison of angiographic characteristics of coronary artery disease in patients with noninsulin-dependent diabetes mellitus compared with matched nondiabetic control subjects. Am J Cardiol 80: 550-556.
35. Horimoto M, Hasegawa A, Ozaki T, Takenaka T, Igarashi K, Inoue H, 2005 Independent predictors of the severity of angiographic coronary atherosclerosis: the lack of association between impaired glucose tolerance and stenosis severity. Atherosclerosis 182: 113-119.
36. Carr MC, 2003 The emergence of the metabolic syndrome with menopause. J Clin Endocrinol Metab 88: 2404-2411.
37. Toth MJ, Sites CK, Eltabbakh GH, Poehlman ET, 2000 Effect of menopausal status on insulin-stimulated glucose disposal: comparison of middle-aged premenopausal and early postmenopausal women. Diabetes Care 23: 801-806.
38. Peppa M, Uribarri J, Vlassara H, 2002 Advanced glycoxidation. A new risk factor for cardiovascular disease? Cardiovasc Toxicol 2: 275-287.
39. Kalish GM, Barrett-Connor E, Laughlin GA, Gulanski BI, 2003 Association of endogenous sex hormones and insulin resistance among postmenopausal women: results from the Postmenopausal Estrogen/Progestin Intervention Trial. J Clin Endocrinol Metab 88: 1646-1652.
40. Reinecke H, Bogdanski J, Woltering A, et al, 2002 Relation of serum levels of sex hormone binding globulin to coronary heart disease in postmenopausal women. Am J Cardiol 90: 364-368.
41. Lapidus L, Lindstedt G, Lundberg PA, Bengtsson C, Gredmark T, 1986 Concentrations of sex-hormone binding globulin and corticosteroid binding globulin in serum in relation to cardiovascular risk factors and to 12-year incidence of cardiovascular disease and overall mortality in postmenopausal women. Clin Chem 32: 146-152.
42. Lindstedt G, Lundberg PA, Lapidus L, Lundgren H, Bengtsson C, Bjorntorp P, 1991 Low sex-hormone-binding globulin concentration as independent risk factor for development of NIDDM. 12-yr follow-up of population study of women in Gothenburg, Sweden. Diabetes 40: 123-128.
43. Sutton-Tyrrell K, Wildman RP, Matthews KA, et al, 2005 Sex-hormone-binding globulin and the free androgen index are related to cardiovascular risk factors in multiethnic premenopausal and perimenopausal women enrolled in the Study of Women Across the Nation (SWAN). Circulation 111: 1242-1249.
44. Heald AH, Anderson SG, Ivison F, et al, 2005 Low sex hormone binding globulin is a potential marker for the metabolic syndrome in different ethnic groups. Exp Clin Endocrinol Diabetes 113: 522-528.
45. Nestler JE, 1993 Sex hormone-binding globulin: a marker for hyperinsulinemia and/or insulin resistance? J Clin Endocrinol Metab 76: 273-274.
46. Golden SH, Ding J, Szklo M, Schmidt MI, Duncan BB, Dobs A, 2004 Glucose and insulin components of the metabolic syndrome are associated with hyperandrogenism in postmenopausal women: the atherosclerosis risk in communities study. Am J Epidemiol 160: 540-548.
47. Apridonidze T, Essah PA, Iuorno MJ, Nestler JE, 2005 Prevalence and characteristics of the metabolic syndrome in women with polycystic ovary syndrome. J Clin Endocrinol Metab 90: 1929-1935.
48. Gaede P, Vedel P, Larsen N, Jensen GV, Parving HH, Pedersen O, 2003 Multifactorial intervention and cardiovascular disease in patients with type 2 diabetes. N Engl J Med 348: 383-393.
Address for correspondence:
Katerina Saltiki, Endocrine Unit, Department of Clinical Therapeutics,
Alexandra University Hospital, 11528 Athens, Greece,
Tel.: +30 210 3381393, Fax: +30 210 7774143, e-mail address: saze@otenet.gr
Received 11-11-07, Revised 20-12-07, Accepted 20-01-08