Abstract
OBJECTIVE: To determine if GC (group-specific component globulin) and CYP2R1 genetic variants have an association with serum 25-OHD3 levels, BMD or bone turnover markers in a population of Chinese postmenopausal women.
DESIGN: We randomly selected 1494 postmenopausal women of the Han ethnic group from seven communities in Beijing. BMD was determined by dual energy X-ray absorptiometry; serum bone turnover markers and 25-OHD3 were measured by the automated Roche electrochemiluminescence system; genotypes of GC and CYP2R1 were detected by the TaqMan allelic discrimination assay. Multiple statistic methods were used to test the associations of SNP genotypes and vitamin D levels.
RESULTS: In our sample, 89.6% women had vitamin D deficiency and another 9.8% had vitamin D insufficiency. The variants of rs2298849 (β=0.105, P<0.001) in GC were significantly associated with serum 25-OHD3 levels. Allele G of rs2298849 might be protective for serum 25-OHD3 level. Among the haplotypes of rs222020-rs2298849, CG (β=0.104, P=0.001) corresponded to increasing serum 25-OHD3 concentrations. CYP2R1 polymorphisms showed some significant association with serum β-CTX and P1NP levels.
CONCLUSIONS: We found that GC variants had a significant association with serum 25-OHD3 levels among postmenopausal women of the Han ethnic group in Beijing, while CYP2R1 variants were not found to be significant.
INTRODUCTION
Vitamin D deficiency is a very common health problem. It is reported that worldwide more than 1 billion children and adults are at risk of vitamin D deficiency.1 The situation is likely to be worse in China. According to a large sample (more than 3,000 individuals) cross-sectional study in older adults living in Beijing and Shanghai, the percentage of vitamin D deficiency and insufficiency were 69.2 and 24.4%, respectively.2 Vitamin D deficiency may decrease peak bone mass in young people and increase the risk of osteoporosis, falls and fractures in the elderly and old.3 It also associated with increasing risk of several autoimmune, cardiovascular, infectious and metabolic diseases.1 Although the main cause of vitamin D deficiency is considered to be lack of sunlight exposure and dietary vitamin D intake, family and twin studies have shown that genetic factors might also affect vitamin levels.4-6
The genome-wide association study (GWAS) and other association studies have been carried out to determine the functional genes. Group-specific component globulin (GC), also called vitamin D binding protein (DBP), is a multifunctional protein which can bind to vitamin D, maintain the vitamin D level in the human body and transport vitamin D to target tissues for processing and utilization.7 CYP2R1 is a member of cytochrome P450 families. It can catalyze the 25-hydroxylation of vitamin D with no sex and species differences and shows catalytic activity toward both vitamin D2 and D3.8 Recently, some studies have observed an association of GC and CYP2R1 variants with serum vitamin D concentration in European populations.9-11 However, it remains to be confirmed whether they have similar effects in Chinese populations. Based on the Peking Vertebral Fracture (PK-VF) study, which is a large-scale epidemiologic study with randomly selected postmenopausal women residing in communities in Beijing, we examined whether the association reported for GC (rs222020 and rs2298849) and CYP2R1 (rs12794714, rs10741657, rs1562902 and rs10766197) variants are also present in this sample. We also explored whether these variants have some relationship with bone mineral density (BMD) and bone turnover markers such as β-C-terminal telopeptide of type 1 collagen (β-CTX) and procollagen type I N propeptide (P1NP).
SUBJECTS AND METHODOLOGY
Participants
Biochemistry
We collected a fasting blood sample from all the participants. Serum concentrations of 25-hydroxyvitamin D3 (25-OHD3), β-CTX and P1NP were measured by a fully automated Roche electrochemiluminescence system (E170, Roche Diagnostics, Switzerland) at PUMCH. The detection limit of β-CTX, P1NP and 25-OHD3 was 0.01 ng/ml, 5 ng/ml and 4 ng/ml, respectively. The intraassay and interassay CV were 2% and 3.1% for β-CTX, 2.3% and 1.7% for P1NP and 5.7% and 6.1% for 25-OHD3.
BMD measurement
Details of BMD measurement have been described previously.12 Briefly, we measured the BMD of the lumbar spine (L2-4), femoral neck (FN) and total hip by dual energy X-ray absorptiometry (DXA).
Genotyping
We searched for single nucleotide polymorphisms (SNP) information on the GC and CYP2R1 genes on the HapMap website (http://hapmap.ncbi.nlm.nih.gov/). SNPs were selected according to the following criteria: 1) high heterozygosity, which means minor allele frequency (MAF) higher than 20% in the Chinese population (CHB in HapMap); 2) classified as tag SNPs; 3) reported to be associated with serum 25-OHD3 level in previous studies.9-11 Finally, we selected two SNPs of the GC gene (rs222020, rs2298849) and four SNPs of the CYP2R1 gene (rs12794714, rs10741657, rs1562902, rs10766197). Each participant was genotyped for all six SNPs by TaqMan allelic discrimination assay (Applied Biosystems, USA). The whole reacting volume was 6μl, including 3μl (approximately 15 ng) sample DNA, 2.5μl TaqMan Universal PCR Master Mix, 0.0875μl TaqMan probe assay and 0.4125μl ddH2O. Reactions were performed on a Real-Time PCR system of ABI Prism 7900 (Applied Biosystems, USA) in a 384-well reaction plate under standard conditions. The genotyping success rate for six SNPs was higher than 98%, and the concordance rate was about 94.4% based on 3% duplicated samples. Genotype frequencies of the six SNPs were similar to those in HapMap-CHB (http://hapmap.ncbi.nlm.nih.gov/) and they were all in Hardy-Weinberg equilibrium.
The Hardy-Weinberg equilibrium (HWE) was tested using the goodness-of-fit chi-square test of the Haploview 4.2 program, which was also used to calculate normalized LD coefficient (D’), and the Pearson correlation coefficient (r2) to measure linkage disequilibrium (LD) between pairwise SNPs. Haplotypes of each participant were determined by Phase 2.02. Serum 25-OHD3 levels were square-root transformed to approximate normality. The independent samples t-test was performed to test the difference in serum 25-OHD3 levels between different seasons. To establish the potential effect of numeric covariates on serum 25-OHD3 levels, a Pearson correlation was conducted as an exploratory analysis (SPSS, version 17.0). Age, BMI and blood collecting season (winter-spring defined as 1, or summer defined as 2) were considered as potential covariates in a linear regression model. Association of the square-root-transformed serum 25-OHD3 levels with age, season and all the SNPs and haplotypes were tested by general linear regression using backward elimination in SPSS, respectively. We also conducted the Pearson correlation or general linear regression to test correlation or association of the 6 SNPs with β-CTX, P1NP and BMD of L2-4, femoral neck and total hip. All analyses were carried out under an additive model. P<0.05 was considered statistically significant. The statistical power was calculated by Quanto software (http://hydra.usc.edu/gxe/). Our study achieved a power of more than 80%.
RESULTS
Basic characteristics of participants
Among the 1494 participants, 91 blood samples were collected in winter, while 714 and 689 blood samples were collected in spring and summer, respectively. Since the sample size of the winter group was small and there was no significantly statistical difference between the winter and spring groups after adjusting age and BMI, we divided the 1494 participants into two season groups: summer and winter-spring. The concentrations of serum 25-OHD3 were significantly different between the summer group and the winter-spring group (P=0.001).
Serum 25-OHD3 levels in different seasons
Association of GC and CYP2R1 genetic variants with serum 25-OHD3 levels
To determine the potential effect of numeric covariates on serum 25-OHD3 levels, a Pearson correlation was conducted as an exploratory analysis (SPSS, version 17.0). Except for all the SNPs under the additive model, age, BMI and blood collecting season (winter-spring defined as 1, or summer defined as 2) were considered as potential covariates in a linear regression model. In our study, SNPs rs222020 (r=0.080, P=0.001) and rs2298849 (r=0.095, P<0.001) of the GC gene, age (r=-0.074, P=0.002) and blood collecting season (r=0.079, P=0.001) were found to have significant correlation with serum 25-OHD3 levels. Following this, the association of the square-root-transformed serum 25-OHD3 levels with age, season and all the SNPs were tested by general linear regression using backward elimination in SPSS. The main results of the SNPs genotyping of the GC and the CYP2R1 genes and their association analyses for serum 25-OHD3 levels are shown in Table 3. SNP rs2298849 (β=0.105, P<0.001) of the GC gene was found to have a significant association with serum 25-OHD3 levels. We did not find any significant association between CYP2R1 genetic variants and serum 25-OHD3 levels.
The LD plots of the GC and CYP2R1 genes are depicted in Figure 1. The results of the haplotype association analyses are displayed in Table 4. The two SNPs of the GC gene were in relatively strong LD (D’>0.8) with each other, as were also the four SNPs of the CYP2R1 gene. P values were calculated using general linear regression (SPSS, version 17.0) under an additive model for those haplotypes with more than 1% frequency. Among the haplotypes of rs222020-rs2298849, CG (β=0.104, P=0.001) corresponded to increasing serum 25-OHD3 concentrations. We did not find any significant association between haplotypes of the CYP2R1 gene and serum 25-OHD3 levels.
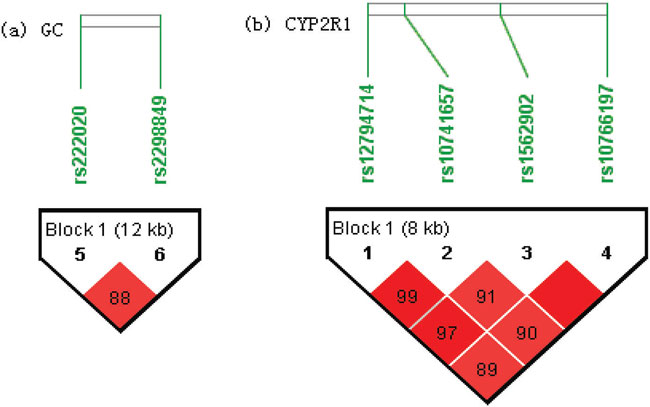
Figure 1. LD plots with D’ values of the GC (a) and CYP2R1 (b) genes in our 1494 participants. The Figure was generated by Haploview. Block definition followed the rules of solid spine of LD. D’ values multiplied by 100 are shown as a number in the diamonds.
Association of GC and CYP2R1 genetic variants with bone turnover markers and BMD
Basic information concerning P1NP, β-CTX and BMD of the lumbar spine (L2-4), femoral neck (FN) and total hip (TH) is shown in Table 1. Some participants failed to attend the BMD test, therefore the available data of BMD was limited, especially as regards BMD of TH.
To determine the potential effect of numeric covariates on bone turnover markers and BMD, a Pearson correlation was conducted (SPSS, version 17.0) as an exploratory analysis. Age and BMI were considered as potential covariates.
As shown in Table 5, in our research SNP rs12794714 (r=-0.053, P=0.021) and BMI (r=-0.052, P=0.022) were found to have significant correlation with β-CTX levels; SNPs rs12794714 (r=-0.064, P=0.007) and rs10741657 (r=0.046, P=0.036) had significant correlation with P1NP levels. In the final linear regression model, SNPs rs12794714 (β=-0.056, P=0.002) and rs10766197 (β=0.044, P=0.012) had statistically significant association with β-CTX levels; SNP rs12794714 (β=-0.017, P=0.014) had statistically significant association with P1NP levels. Although we found some statistically significant results, as the magnitude of association was rather small, we did not do further haplotype association analysis.
As shown in Table 6, in our research, age (r=-0.299, P<0.001) and BMI (r=0.183, P<0.001) had significant correlation with BMD of L2-4; age (r=-0.521, P<0.001) and BMI (r=0.210, P<0.001) had significant correlation with BMD of FN; age (r=-0.535, P<0.001) and BMI (r=0.272, P<0.001) had significant correlation with BMD of TH. None of the 6 SNPs were found to have significant correlation with BMD of L2-4, FN, or TH (P>0.05). We thus did not proceed to further linear regression to test the association between SNPs or haplotypes and BMD.
We found that the variants of rs2298849 (β=0.105, P<0.001) in the GC gene were significantly associated with serum 25-OHD3 levels. Allele G of rs2298849 might be protective for serum 25-OHD3 levels. The variants of rs222020 (r=0.080, P=0.001) in the GC gene were correlated with serum 25-OHD3 levels, although they were not significantly associated with serum 25-OHD3 levels in the final linear regression model. This finding is consistent with the study by Bu et al.11 Previous studies on the GC gene focused more on functional SNP rs4588 located in the exon. Its A/Lyn allele corresponds to the GC2 protein, which was found to be significantly associated with decreased serum 25-OHD3 level7,14,15 Lu et al. observed that the variants of rs4588 were associated with plasma 25-OHD3 levels in a Chinese population,16 while Ahn et al. and Wang et al. found that rs2282679 variants were associated with serum 25-OHD3 levels in white Europeans.9,10 In the HapMap Data, rs2282679 and rs4588 are in complete LD (r2=1) in CEU and CHB populations, while these two SNPs are both in very weak LD with rs222020 and rs2298849 (r2 ranging from 0.08 to 0.15). As rs222020 and rs2298849 are both located in the intron, there might be some new functional site which is independent of rs4588 and can influence serum 25-OHD3 levels. Fine genotyping in the area near rs222020 and rs2298849 might need to be done to discover new functional sites.
We detected no significant association between CYP2R1 polymorphisms and serum 25-OHD3 levels in Chinese postmenopausal women. This finding is consistent with the previous study in a Chinese population by Lu et al.16 However, CYP2R1 genetic variants were found to be associated with vitamin D level in many studies in white Europeans.9-11 Since we had >99% power to find the beta values reported by Bu et al. in white Europeans,11 the discrepancy with previous studies may result from LD pattern variation of the causal SNP and the four SNPs detected in our study between Chinese Hans and white Europeans. Previous studies have hypothesized that genetic factors may play a much more important role in influencing vitamin D level in men than in women.4 As we had only female participants, our results may not represent the status of men. Another possibility is that other vitamin D 25-hydroxylation enzymes play a more important role in Chinese Hans than in white Europeans, thus CYP2R1 polymorphisms would hypothetically not influence serum 25-OHD3 level in Chinese Hans.
In our study, we found CYP2R1 polymorphisms to be significantly associated with two bone turnover markers: β-CTX and P1NP. As we discovered no significant association between CYP2R1 polymorphisms and serum 25-OHD3 levels in our study, the CYP2R1 gene might influence these two bone turnover markers via some as yet unknown mechanism. Since we did not locate any previous studies on the association between CYP2R1 genetic polymorphisms and the two bone turnover markers, this result may need to be further corroborated considering the small magnitude of the association.
CONCLUSIONS
In conclusion, we found that GC genetic variants had a significant association with serum 25-OHD3 level among postmenopausal women of the Han ethnic group in Beijing, while CYP2R1 genetic variants were not observed as being significant. Association of CYP2R1 genetic variants with β-CTX and P1NP needs to be further substantiated.
GRANTS SUPPORTS
This research was supported by the National Natural Science Foundation of China (No.81070687 and 81170805), National Science and Technology Pillar Program (2006BAI02B03), National Science and Technology Major Projects for “Major New Drugs Innovation and Development” (Grant2008ZX09312-016), Beijing Natural Science Foundation (No. 7121012) and Scientific Research Foundation of Beijing Medical Development (No. 2007-3029).
REFERENCES
1. Holick MF, 2011 Vitamin D deficiency in 2010: health benefits of vitamin D and sunlight: a D-bate. Nat Rev Endocrinol 7: 73-75.
2. Lu L, Yu Z, Pan A, et al, 2009 Plasma 25-hydroxyvitamin D concentration and metabolic syndrome among middle-aged and elderly Chinese individuals. Diabetes Care 32: 1278-1283.
3. Holick MF, 2011 Vitamin D: a d-lightful solution for health. J Investig Med 59: 872-880.
4. Arguelles LM, Langman CB, Ariza AJ, et al, 2009 Heritability and environmental factors affecting vitamin D status in rural Chinese adolescent twins. J Clin Endocrinol Metab 94: 3273-3281.
5. Shea MK, Benjamin EJ, Dupuis J, et al, 2009 Genetic and non-genetic correlates of vitamins K and D. Eur J Clin Nutr 63: 458-464.
6. Snellman G, Melhus H, Gedeborg R, et al, 2009 Seasonal genetic influence on serum 25-hydroxyvitamin D levels: a twin study. PLoS One 4: e7747.
7. Fang Y, van Meurs JB, Arp P, et al, 2009 Vitamin D binding protein genotype and osteoporosis. Calcif Tissue Int 85: 85-93.
8. Strushkevich N, Usanov SA, Plotnikov AN, Jones G, Park HW, 2008 Structural analysis of CYP2R1 in complex with vitamin D3. J Mol Biol 380: 95-106.
9. Ahn J, Yu K, Stolzenberg-Solomon R, et al, 2010 Genome-wide association study of circulating vitamin D levels. Hum Mol Genet 19: 2739-2745.
10. Wang TJ, Zhang F, Richards JB, et al, 2010 Common genetic determinants of vitamin D insufficiency: a genome-wide association study. Lancet 376: 180-188.
11. Bu FX, Armas L, Lappe J, et al, 2010 Comprehensive association analysis of nine candidate genes with serum 25-hydroxy vitamin D levels among healthy Caucasian subjects. Hum Genet 128: 549-56.
12. Zhao J, Xia W, Nie M, et al, 2012 A haplotype of MATN3 is associated with vertebral fracture in Chinese postmenopausal women: Peking Vertebral Fracture (PK-VF) study. Bone 50: 917-924.
13. Purcell S, Neale B, Todd-Brown K, et al, 2007 PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81: 559-575.
14. Abbas S, Linseisen J, Slanger T, et al, 2008 The Gc2 allele of the vitamin D binding protein is associated with a decreased postmenopausal breast cancer risk, independent of the vitamin D status. Cancer Epidemiol Biomarkers Prev 17: 1339-1343.
15. Lauridsen AL, Vestergaard P, Hermann AP, et al, 2005 Plasma concentrations of 25-hydroxy-vitamin D and 1,25-dihydroxy-vitamin D are related to the phenotype of Gc (vitamin D-binding protein): a cross-sectional study on 595 early postmenopausal women. Calcif Tissue Int 77: 15-22.
16. Lu L, Sheng H, Li H, et al, 2012 Associations between common variants in GC and DHCR7/NADSYN1 and vitamin D concentration in Chinese Hans. Hum Genet 131: 505-512
Address for correspondence:
Prof. Weibo Xia, Department of Endocrinology, Key Laboratory of Endocrinology, Ministry of Health, Peking Union Medical
College Hospital, Chinese Academy of Medical Science, Beijing 100730 China, Tel.: 86-10-6915-5358, Fax: 86-10-6915-5358,
E-mail: xiaweibo@medmail.com.cn
Received 23-05-2013, Accepted 08-09-2013