Abstract
OBJECTIVE:The aim of this study was to evaluate the changes in the levels of vasoactive eicosanoid hormone-like substances PGE2, PGI2 and TXA2 in hemodialysis (HD)patients who were following a long-term physical training program during the hemodialysis session. DESIGN: A total of 50 patients with Chronic Kidney Disease (CKD) (stage 5)on hemodialysis and 35 healthy individuals who served as controls (C) were evaluated. The 50 CKD patients were divided into two groups: the HD group consisted of 31 patients who received usual care without any physical activity during the hemodialysis sessions, while group HD/Exer included 19 patients who followed a program of physical exercise for six months. Plasma levels of PGE2, 6-Keto-PGF1(the stable derivative of PGI2) and TXB2 (the stable derivative of TXA2) were measured by reliable enzymo-immunoassay methods (EIA) in HD and HD/Exer patients before and after the hemodialysis sessions as well as in the group of C. RESULTS: The plasma levels of PGE2 and 6-keto-PGF in group HD Exer/before patients were higher than those in group HDbefore (20.39±5.82 and 1449.19±553.41 vs 17.68±5.36 and 1295.10±384.43 pg/ml, p=0.044 and p=0.067, respectively), while the plasma levels of TXB2 were lower in HD Exer/before patients compared to HDbefore(499.76±67.51 vs 608.01±80.23 pg/ml, p=0.041). The plasma levels of PGE2 and 6-keto-PGF1αin group HD Exer/after patients were significantly higher compared to those in HDafter patients (23.01±5.70 and 1618.19±435.07 vs 16.57±4.97 and 1005.44±317.16 pg/ml, p<0.001 and p<0.040, respectively). However, significantly lower values in the plasma levels of TXB2 in HD Exer/after compared to HDafter patients (363.10±51.91 vs 439.75±62.34 pg/ml, p=0.030) were detected. As expected, PGE2 and 6-keto-PGF1α values were lower in C than in the groups of patients with CKD. CONCLUSIONS: The data indicate that exercise training during HD exerts a beneficial effect on the levels of the vasoactive eicosanoid hormone-like substances in patients on HD.
Eicosanoid hormone-like substances are produced from arachidonic acid (a.a.) via the action of cyclo-oxygenases and synthases. The main eicosanoid hormone-like substances are: Prostaglandin Ε2 (PGE2), Prostacyclin Ι2 (PGI2) and Thromboxane A2 (TXA2). Arachidonic acid is released from the cellular membranes of human cells (platelets, macrophages, glomerular mesangial cells, others) by the action of the phospholipase A2. Arachidonic acid is converted to prostaglandin endoperoxides by cyclo-oxygenase (endoperoxide synthase). Endoperoxides are subsequently converted to prostaglandins (PGs), prostacyclin (PGI2) and thromboxane A2 (TXA2).1-5
Prostaglandin Ε2 (PGE2) is a vasodilative local hormone which belongs to the family of eicosanoid hormone-like substances. The cells that mainly produce PGE2 are the epithelial cells, the fibroblasts, the monocytes-macrophages and the eosinophils. Recently, a different metabolic pathway of PGE2synthesis was disclosed. Thus, in cases of intense oxidative stress, when 15-F2t-isoprostane (15-F2t-IsoP) production is increased, prostaglandin E2 is produced at high rates (15%-40%) by 15-F2t– IsoP epimerization. PGE2 has significant effects on several organs, including the kidneys. It has been found that PGE2 induces a rise in the renal blood flow, in diuresis, natriuresis and kaliuresis. Additionally, PGE2 increases the Glomerular Filtration Rate (GFR), inhibits the reabsorption of chloride ions (Cl–) and results in increased renin secretion and erythropoeitin release.1,2,6-14
Prostacyclin Ι2 (PGI2) is another local hormone which belongs to the family of eicosanoid hormone-like substances. PGI2 has a powerful vasodilative action, inhibits the accumulation of platelets and obstructs the vasocostrictive action of thromboxane Α2. Additionally, it has an antithrombotic action in the vessel intima; in the kidneys, the action of PGI2 is similar to that of PGE2. The cells that mainly produce PGI2 are the endothelial cells, smooth muscle cells, macrophages and fibroblasts. PGI2 is rapidly converted to 6-Keto-PGF1α, a chemically stable but biologically inactive product.1,2,15-18
Thromboxane A2 (TXA2) is a catabolic product of arachidonic acid. Besides this, TXA2 is produced in vivo and non-enzymatically by free radical-catalyzed lipid peroxidation via isoprostane endoperoxides re-arrangement. Being a very efficient vasoconstrictor agent and inducing a potent platelet aggregation, TXA2 is an antagonist to prostacyclin. It is considered that physiological balance between the two components plays an important regulatory role in the maintenance of normal vascular tone and in the pathogenesis of various cardiovascular disorders. As regards the kidneys, TXA2 causes marked renal vasoconstriction; consequently, enhanced renal TXA2 production reduces renal blood flow and glomerular filtration rate. Since TXA2 is rapidly converted to thromboxane B2 (TXB2), a chemically stable but biologically inactive hydration product, thromboxane synthesis by biological tissues has been monitored by measuring TΧΒ2.1,2,19-21
Enhanced eicosanoid hormone production has been detected in a number of diseases such as cardiovascular disorders, diabetes mellitus and inflammatory, immunological and renal diseases.7,8,21-29 Physical exercise induces the release of vasodilative prostaglandins (PGE2 and PGI2) by the skeletal muscles; consequently, these substances may contribute to arterioles vasodilation and hyperhemia, which occurs in the skeletal muscles during physical exercise. Plasma levels of TXB2 decrease during short- or long-term exercise. Some researchers, however, have reported a rise in thromboxane plasma levels during physical exercise.30-33 We have shown that the interstitial and plasma levels of PGE2 and 6-keto-PGF1a also increase during exercise.9,34,35 There are no available data with regard to changes of the above-mentioned bioactive eicosanoid hormone-like substances (PGE2, PGI2 and TXA2) in hemodialysis patients who participate in a program of physical training during the hemodialysis session.
The aim of the present study was to evaluate the effect of physical exercise during the hemodialysis session on the levels of vasoactive eicosanoid hormones, derivatives of arachidonic acid, in patients undergoing hemodialysis and participating in a program of physical exercise during the hemodialysis session.
SUBJECTS AND METHODOLOGY
The study group included 35 healthy individuals who served as controls (group C), 22 men and 13 women, aged 56.7±13.8 (mean±SD) years, and 50 patients with end-stage renal failure (stage 5) on chronic hemodialysis (HD). All our study subjects were initially evaluated at the Renal Dialysis Unit of the 1st Department of Internal Medicine at AHEPA University Hospital, Thessaloniki, Greece, where detailed medical history was recorded and careful clinical examination was performed. Patients with acute or chronic inflammatory diseases, a history of myocardial infarction, unstable angina or stroke in the past six months, a history of malignancy, as well as patients receiving cyclooxygenase inhibitors or vitamin E and other supplements, were excluded from the study.
The study was conducted in accordance with the Declaration of Helsinki (1985 amendment) and was approved by the Ethics Committee of the School of Medicine, Aristotle University of Thessaloniki, Greece. Informed written consent was obtained from all subjects included in this study prior to study initiation.
The hemodialysis patients were divided into two groups: HD patients who were not following a physical activity program and received usual care and HD patients performing an exercise program (HD/Exer). Each group (HD and HD/Exer) was studied before HD (HDbefore and HD Exer/before, respectively) and after HD (HDafter and HD Exer/after respectively). Subgroup HDbefore included 31 patients (21 men and 10 women), aged 58.9±15.8 years, with an estimated GFR <15.0ml/min/1.73 m2, who had been on chronic periodic hemodialysis for a period of 64.8±12.1 months. The hemodialysis procedure took place three times a week with polysulphone filters and lasted four hours. Subgroup HDafter included the same patients after the hemodialysis session. Subgroup HD Exer/before comprised 19 patients before the hemodialysis session, with the same estimated GFR (12 men and 7 women), aged 58.1±8.7 years, who had been on hemodialysis for a period of 91.2±15.3 months; they participated voluntarily in a program of mild physical exercise carried out during hemodialysis for a time period of six months prior to entrance to the study. Subgroup HD Exer/after comprised the same patients as subgroup HD Exer/before and were studied after the HD. All HD/Exer patients underwent thorough examination of their cardiovascular and respiratory system; patients with severe cardiac arrhythmias (stage ≥ΙΙΙ according to Lown), unsteady coronary disease, congestive heart failure (stage >ΙΙ according to the Νew York Heart Association), recent myocardial infraction, valvulitis or chronic respiratory failure were excluded from the study. In addition, patients suffering from resistant hypertension and severe hyperparathyroidism (Ι-ΡΤΗ >600 pg/ml), and individuals with osteodystrophy or severe anemia (Ηb <10 g/dl), were also excluded from the study.
Information on subject demographics, medical history, concomitant medication, smoking, dietary habits, anthropometric measurements, blood pressure measurements and heart rate were recorded according to a standardized protocol. Blood samples from the healthy control subjects were drawn in the morning after a 12-hour overnight fast in order to determine routine hematologic and biochemical parameters, as well as the parameters under investigation. In the hemodialysed patients, blood samples were drawn under the same conditions before and after the hemodialysis session. Estimation of glomerular filtration rate was performed using the modification of Diet in Renal Disease study formula,36 according to the guidelines.37
Exercise Training Rehabilitation Program during hemodialysis
Patients of group HD/Exer followed a 6-month rehabilitation program during the first two hours of their hemodialysis sessions under the supervision of two physical physiologists. Patients participated in the physical exercise program three times a week for 60 minutes each time. Special stationary bicycles (type Motomed Letto 713/W 1498)were moved up to the dialysis chairs or beds. Patients cycled for 30 to 60 min and afterwards performed exercise for strength and flexibility for 15 to 30 min. The first five minutes consisted of warm-up, the second period consisted of training at desired workload (cycling and exercises) and the last five minutes consisted of cool-down. Exercise intensity corresponded constantly to 13 (somewhat vigorous) of the Borg perceived exertion scale.35-38
Biochemical – Endocrine Parameters
Plasma PGE2 was determined by a competitive enzymo-immunoassay method (ΕΙΑ), using kits from Cayman. The quantification of 6-keto-PGF1α and TXB2 was also performed by an ΕΙΑ method using kits from R and D.
The normal plasma levels according to the PGE2 quantification method were 3-12 pg/ml with intra-assay coefficient of variation (CV) 5.1% and inter-assay CV 5.8%. The sensitivity of the method used to determine 6-keto-PGF1α in the plasma was <5 pg/ml, whereas the intra-assay coefficient of variation (CV) was 5% and the inter-assay CV 7.3%.
The performance characteristics of the TXB2 method were as follows: sensitivity 8 pg/ml and intra- and inter-assay coefficients of variation (CV) 3.6% and 7.7%, respectively.
Statistical Analysis
Statistical analysis was performed using the SPSS version 13 software (SPSS Inc., Chicago, III, USA) for Windows XP. Continuous variables are expressed as mean ± standard deviation (mean ± SD). Comparisons of the categorical variables between the groups was performed by the x2 test or Fisher’s exact test whenever the marginal expected count was <5; to compare the continuous variables we used the one-way analysis of variance (ANOVA). In order to explore the differences among various pairs of the four groups studied, we performed Student’s tests for independent variables with Bonferroni correction for multiple testing. In addition, we performed simple linear regression analysis and calculated Pearson’s correlation coefficients so as to explore possible relations between estimated GFR and the parameters under study. A value of p<0.05 (two-tailed) was considered statistically significant.
RESULTS
Basic demographic data as well as the clinical characteristics of the studied subjects are presented in Table 1 . There were no statistically significant differences among the groups with regard to sex, age, body mass index (BMI) and proportion of smokers. When comparing the HD groups, no significant differences in the proportion of patients with a history of hypertension, diabetes and cardiovascular disease were noticed.
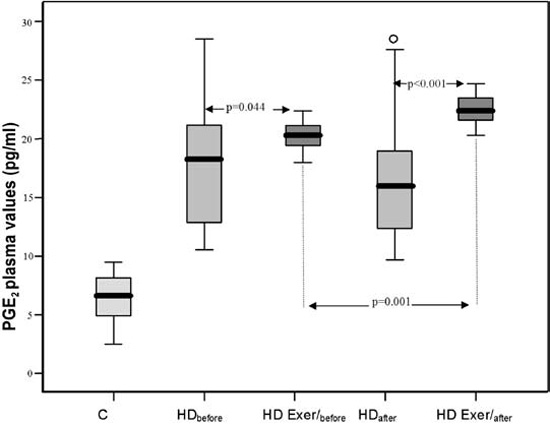
a) Before hemodialysis, the PGE2 plasma levels of the patients who underwent a physical training program during the hemodialysis session (Subgroup HDExer/before) were significantly higher than the PGE2 plasma levels of controls (p<0.001) and of hemodialysis patients who did not participate in the physical training program (Subgroup HDbefore, p=0.044).
After hemodialysis, the plasma PGE2 levels in subgroup HD Exer/after were much higher than the PGE2 plasma levels in subgroup HD Exer/before (p=0.001), as well as the levels in subgroup HDafter (p<0.001) (Table 2 , Figure 1A).
HD: Hemodialysis
Exer: Exercise
PGE2: Prostaglandin E2
6-keto-PGF1α: 6-keto prostaglandin F1α
TXB2: Thromboxane B2
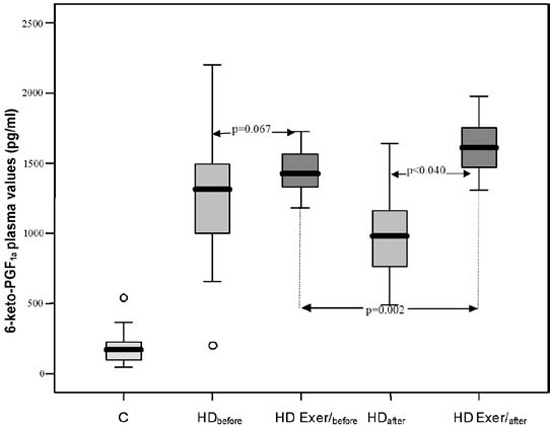
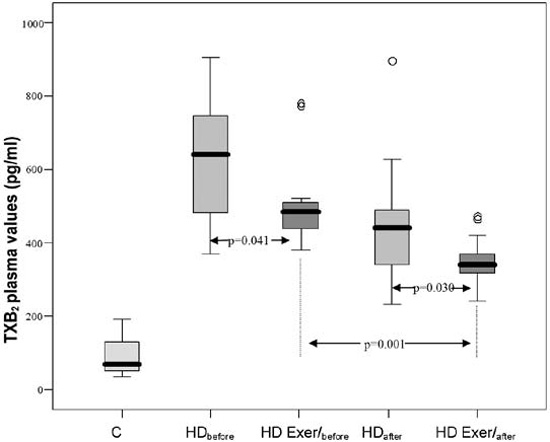
Figure 1. Mean plasma values of PGE2 (A), 6-keto-PGF1α (B) and TXB2 (C) in controls and in the various subgroups HDbefore, HD Exer/before, HDafter and HD Exer/after. Comparisons C vs all the other groups, p<0.001.
b) The 6-keto-PGF1α plasma levels in patients before the hemodialysis in subgroup HD Exer/before were higher but not significantly (p=0.067) compared to the 6-keto-PGF1α plasma levels in the respective subgroup HDbefore and significantly higher than the corresponding levels observed in controls (p<0.001).
The plasma levels of 6-keto-PGF1α in subgroup HD Exer/after were significantly higher than the 6-keto-PGF1α plasma levels in subgroup HD Exer/before (p=0.002), as well as of the patients in subgroup HDafter (p<0.040) (Table 2, Figure 1B). Furthermore, there was a positive correlation between PGE2 and 6-keto-PGF1α values in the patients of groups HD Exer/before and HD Exer/after (r=0.94, p<0.001, and r=0.99, p<0.001, respectively; Figures 2A and 2B).
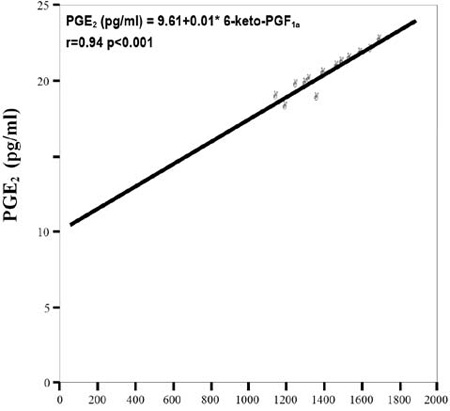
Figure 2A. Correlation between PGE2 and 6-keto-PGF1α in the group HD Exer/before.
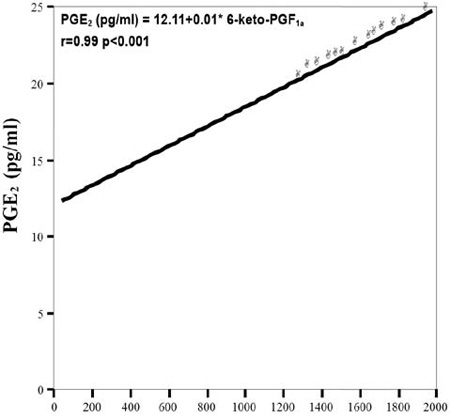
Figure 2B. Correlation between PGE2 and 6-keto-PGF1α in the group HD Exer/after.
c) The TXB2 plasma levels of the patients in subgroup HDExer/before were significantly lower than the TXB2 plasma levels in the respective patients of subgroup HDbefore (p=0.041) and significantly higher than the TXB2 plasma levels of controls (Group C, p<0.001).
After hemodialysis, the TXB2 plasma levels in patients of subgroup HD Exer/after were significantly lower than the TXB2 plasma levels in patients of subgroup HDafter (p= 0.030).
The plasma levels of TXB2 in patients after hemodialysis (Subgroup HD Exer/after) were much lower than in the same patients subgroup HD Exer/before (p=0.001) (Table 2, Figure 1C).
The ratio TXB2/PGE2 in groups C, HDbefore, HD Exer/before, HDafter,as calculated according to the values of Table 2, is 13.3 :1, 34.3 :1, 21.5 :1 and 26.5 :1, respectively. This ratio of group HD Exer/after almost attains the ratio of healthy individuals (15.8:1). Similarly, the ratio 6-keto-PGF1α/TXB2 in groups C, HDbefore,HDExer/before, HDafter was 3.9:1, 2.1:1, 3.2:1 and 2.2:1, respectively. This ratio of group HD Exer/after almost reaches the values of healthy individuals (4.4 :1).
DISCUSSION
There are no literature data evaluating the potential benefit of physical exercise applied during hemodialysis on the levels of vasoactive eicosanoid hormone-like substances.
In the present study, higher values of PGE2 in the plasma of hemodialysis patients who participated in the mild physical exercise program for a long time were detected compared to values of HD patients not participating in the exercise program. It is noteworthy that the ratios TXB2/PGE2 and 6-keto-PGF1α/TXB2, which were found to be abnormal in HD patients, improved after physical training during the HD session and almost reached the ratios of healthy individuals. In previous studies investigating PGE2 plasma changes in healthy individuals after vigorous physical training for a short period of time, we found that the PGE2 plasma levels rise significantly. However, high intensity physical training cannot be applied in hemodialysis patients because it is dangerous for them.7-9,34
The increase of PGE2 plasma levels in hemodialysis patients who participated in a mild physical training program for a long period of time, as compared to those who did not undergo such a training program, could be related to certain alterations known to occur in HD patients on an exercise program. Thus, morphological, functional and metabolic adjustments of the skeletal muscles are improved in hemodialysis patients participating in such programs of physical training during hemodialysis.38-40
The rehabilitation of the hemodialysis patients is enhanced, most likely because aerobic exercise induces elongation and an increase in the diameter of the striated muscle fibre, improves their capillary vasculature, as well as their aerobic capacity, and positively affects their blood pressure measurements, their brain function and the lipid profile.38,39
The increased ionic calcium of the cell sarcoplasma in the skeletal muscles, which is prevalent during the muscle contractions, possibly stimulates muscle cells phosphorylase Α2; the outcome is an increased production of arachidonic acid, with subsequent increased synthesis of ΡGE2.31,41
Bradykinin production is increased during physical training, resulting in activation of phospholipase Α2 with further increase in the biosynthesis of PGE2.42
It should be pointed out that the biosynthesis of PGE2 in the hemodialysis patients who participate in programs of physical exercise during the hemodialysis session could be even higher than the rates we found, since physical exercise induces an increased PGE2 clearance via the beneficial morphological, metabolic and functional changes in the muscles involved.
The increased 6-Keto-PGF1α plasma levels before and particularly after hemodialysis in hemodialysis patients who participated in the program of mild physical exercise for a long period of time could be attributed to the same reasons that also caused the increase of PGE2.42 It should be emphasized that in previous studies an analogous increase of 6-keto-PGF1α in biological fluids after intense physical training was detected.9,35 It seems that the favourable biochemical-endocrine adjustments which are observed as a result of brief intense physical exercise in healthy individuals are also observed in hemodialysis patients who exercise mildly but for a longer period of time.
The decrease of plasma TXB2 levels in patients on hemodialysis following a program of physical training could be attributed to morphological, metabolic and functional changes in the skeletal muscles, like those observed during similar training programs, as well as to the decrease of platelet activation during exercise.38-40 This finding is similar to that of previous studies, in which a significant decrease of TXA2 was detected in healthy humans during dynamic contractions.9,34 These changes in the eicosanoid hormone-like substances in hemodialysis patients may contribute to the vasodilation and hyperemia that occur in the working muscles during exercise.30 We would like to point out that the favourable alterations in the plasma levels of PGE2, 6-keto-PGF1α and TXB2 in hemodialysis patients detected in the present study have been demonstrated in healthy subjects during exercise.5,30,34,35 Nevertheless, the present findings in HD patients constitute novel observations.
It should be briefly mentioned that patients with CKD, whether undertaking HD or not, experience multiple benefits from physical exercise; resistance exercise training program in patients with severe CKD was found to cause a significant increase in the size of forearm veins.43 In hemodialysis patients, handgrip training and intermittent compression of the upper arm veins led to increased diameter of forearm arteries and veins and improved endothelium-dependent vasodilation.44 Moreover, three months of aerobic exercise training was found to improve arterial stiffness in patients on hemodialysis.45 It was also suggested that a low-intensity exercise program during HD increased the rate of urea removal, perhaps due to the acute increases in blood flow to working muscles and reduction in oxidative stress.7,8,46 There is also evidence that exercise during hemodialysis can be used as an adjunctive therapy to enhance dialysis efficacy.46,48
Our data support the view that the plasma levels of the vasodilative eicosanoid hormones, derivatives of arachidonic acid (prostaglandin E2 and prostacyclin I2), in patients on hemodialysis who follow a program of physical training carried out during the hemodialysis session were increased, as compared to those in patients on hemodialysis who were not following a program of physical training. By contrast, the plasma levels of vasoconstrictive thromboxane B2 were simultaneously reduced. These alterations suggest that an exercise program of moderate or low intensity during HD, apart from being safe, is also substantially beneficial in many respects since it improves their vasoactive hormonal profile. Consequently, such an exercise program can be used as an adjunctive therapy to enhance dialysis efficacy.
REFERENCES
1. Smyth EM, Burne A, Fitzgerald GA 2006 Lipid-derived autacoids: Eicosanoids and platelet-activating factor. In handbook of the pharmacological basis of therapeutics. (Goodman and Gilman’s), (eds) McGraw-Hill, New York, pp, 653-670.
2. Smyth EM, Fitzegerald GA 2003 Prostaglandin mediator. In handbook of cell signaling (Bradshaw, R.D.), (ed.) Academic Press, San Diego, pp, 265-273.
3. Malkowski MG, Ginell SL, Smith WL, Garavito RM, 2000 The productive conformation of arachidonic acid, bound to prostaglandin synthase. Science 289: 1933-1937.
4. Smith WL, Langenbach R, 2001 Why there are two cyclooxygenase isozymes. J Clin Invest 107: 1491-1495.
5. Lands WE, 1991 Biosynthesis of prostaglandins. Annu Revu Nutr 11: 41-60.
6. Gao L, Zackert WE, Hasford JJ, et al, 2003 Formation of prostaglandins E2 and D2 via the isoprostane pathway: a mechanism for the generation of bioactive prostaglandins independent of cyclooxygenase. J Biol Chem 278: 28479-28489.
7. Karamouzis I, Sarafidis P, Karamouzis M, et al, 2008 Increase in oxidative stress but not in antioxidant capacity with advancing stages of chronic kidney disease. Am J Nephrol 28: 397-404.
8. Karamouzis I, Grekas D, Karamouzis M, et al, 2008 Enhanced oxidative stress with a gradient between plasma and muscle interstitial fluid in patients with end stage renal failure on hemodialysis. Hormones (Athens) 7: 62-69.
9. Karamouzis M, Langberg H, Kjaer M, B?low J, Saltin B, 1999 A comparative study of the influence of dynamic and static isometric intermittent contractions on the intramuscular accumulation of prostaglandins and thromboxanes. A study with microdialysis. Medical Biochemistry 1: 233-242.
10. Dunn MJ, Hood VL, 1977 Prostaglandins and the Kidney. Am J Physiol 233: 169-184.
11. Strokes JB, 1979 Effect of prostaglandin E2 on chloride transport across the rabbit thick ascending limb of Henle: Selective inhibitions of the medullary portion. J Clin Invest 64: 495-502.
12. Gross DM, Brookins J, Fink GD, Fisher JW, 1976 Effects of prostaglandins A2, E2 and F2α on erythropoietin production. J Pharmacol Exp Ther 198: 489-496.
13. Neisius K, Olsson R, Rukwied R, Lischetzki G, Schmelz M, 2002 Prostaglandin E2 induces vasodilation and pruvitus but no proteins extravasation in atopic dermatitis and controls. J Am Acad Dermatol 47: 28-32.
14. Eleftheriadis E, Kotzampassi K, Tzioufa V, Karamouzis M, Aletras H, 1990 Effects of famotidine on gastric mucus of the rat. Res Exp Med (Berl) 190: 219-222.
15. Hinderliter AL, Willis PW, Barst RI, et al, 1997 Effects of long-term infusion of prostacyclin (epoprostenol) on echocardiographic measures of right ventricular structure and function in primary pulmonary hypertension. Circulation 95: 1479-1486.
16. Kozek–Langenecker SA, Spiss CK, Michalek-Sauberer A, Felfernig M, Zimpfer M, 2003 Effect of prostacyclin on platelets, polymorphonuclear cells and heterotypic cell aggregation during hemofiltration. Crit Care Med 31: 864-868.
17. Hertz R, Berman I, Keppler D, Bar-Tana J, 1996 Activation of gene transcription by prostacyclin analogues is mediated by the peroxisome-proliferators-activated receptor (PPAR). Eur J Biochem 235: 242-247.
18. Horiba N, Kumano E, Watamabe T, et al, 2002 Subtotal nephrectomy stimulates cyclooxygenase 2 expression and prostacyclin synthesis in the rat remnant kidney. Nephron 91: 134-141.
19. Morrow JD, Awad JA, Wu A, Zackert WE, Daniel VC, Roberts LJ, 1996 Nonenzymatic free radical-catalyzed generation of thromboxane-like compounds (isothromboxanes) in vivo. J Biol Chem 271: 23185-23190.
20. Patrono C, Patrignani P, Davi G, 1993 Thromboxane biosynthesis and metabolism in cardiovascular and renal disease. J Lipid Mediat 6: 441-451.
21. Thomas DW, Coffman TM, 1998 A genetic approach for studying the role of thromboxane A2 in the kidney. Kidney Int Suppl 67: 84-87.
22. Minuz P, Patrignani P, Caino S, et al, 2004 Determinants of platelet activation in human essential hypertension. Hypertension 43: 64-70.
23. Zou MH, Leist M, Ullrich V, 1999 Selective nitration of prostacyclin synthase and defective vasorelaxation in atherosclerotic bovine coronary arteries. Am J Pathol 154: 1359-1365.
24. Kyrle PA, Stockenhuber F, Brenner B, et al, 1998 Evidence for an increased generation of prostacyclin in the microvasculature and an impairment of the platelet-alpha granule release in chronic renal failure. Thromb Haemost 60: 205-208.
25. Strandvik B, Svensson E, Seyberth HW, 1996 Prostanoid biosynthesis in patients with cyctic fibrosis. Prostaglandins Leucot Essent Fatty Acids 55: 419-425.
26. Karamouzis M, Langberg H, Skovgaard D, Bülow J, Kjaer M, Saltin B, 2001 In situ microdialysis of intramuscular prostaglandin and thromboxane in contracting skeletal muscle in humans. Acta Physiol Scand 171: 71-76.
27. Langberg H, Skovgaard D, Karamouzis Μ, Bülow J, Kjaer M, 1999 Metabolism and inflammatory mediators in the peritendinous space measured by microdialysis during intermittent isometric exercise in humans. J Physiol 515: 919-927.
28. Minuz P, Patrignani P, Gaino S, et al, 2002 Increased oxidative stress and platelet activation in patients with hypertension and renovascular disease. Circulation 106: 2800-2805.
29. Kitagawa M, Tanigawa K, Iwata M, 1999 The role of the spleen, especially regarding changes in both thromboxane A2 and the remnant liver dysfunction after extensive hepatetectomy. Surg Today 29: 137-142.
30. Wilson JR, Kapoor SC, 1993 Contribution of prostaglandins to exercise induced vasodilation in humans. Am J Physiol 265: 171-175.
31. Symons JP, Theodossy SJ, Longhurst JC, Stebbins CL, 1991 Intramuscular accumulation of prostaglandins during static contraction of cat triceps surae. J Appl Physiol 71: 1837-1842.
32. Sparks V, Thompson L 1984 Physiology of Blood Circulation. In : Abramson D, Dobrin P, Orlando Fl (eds) Blood Vessels and Lymphatics in Organ System, Ny: Academic Press; pp, 647-652.
33. Todd MK, Goldfarb AH, Burleson C, 1997 Comparison of methods for evaluating exercise-induced changes in thromboxane B2 and beta-thromboglobulin. Med Sci Sports Exerc 29: 700-704.
34. Karamouzis M, Karamouzis I, Vamvakoudis E, et al, 2001 The response of muscle interstitial prostaglandin E2 (PGE2), prostacyclin I2 (PGI2) and thromboxane A2 (TXA2) levels during incremental dynamic exercise in humans determined by in vivo microdialysis. Prostaglandins Leukot Essent Fatty Acids 64: 259-263.
35. Karamouzis I, Sioulis A, Karamouzis M, et al, 2006 Effects of physical training on plasma levels of thromboxane (TXA2) in patients on hemodialysis. Scientific Annals of the Medical school of Thessaloniki 33: 71-75.
36. Levey AS, Bosch JP, Lewis JB, Greene T, Rogers N, Roth D, 1999 A more accurate method to estimate glomerular filtration rate from serum creatinine: a new prediction equation. Modification of Diet in Renal Disease Study Group. Ann Intern Med 130:461-470.
37. National Kidney Foundation, 2002: K/DOQI Clinical Practice guidelines for chronic kidney disease: evaluation, classification and stratification. Kidney Disease Outcome Quality Initiative. Am J Kidney Dis 39: Suppl 1: S1-S266.
38. Konstantinidou E, Koukouvou G, Kouidi E, Deligiannis A, Tourkantonis A, 2002 Exercise training in patients with end-stage renal disease on hemodialysis: comparison of three rehabilitation programs. J Rehabil Med 34: 40-45.
39. Deligiannis A, 2004 Cardiac adaptations following exercise training in hemodialysis patients. Clin Nephrol 61: 539-545.
40. Deligiannis A, 2004 Exercise rehabilitation and skeletal muscle benefits in hemodialysis patients. Clin Nephrol 61: 546-550.
41. Craven PA, Briggs R, DeRubertis FR, 1980 Calcium-dependent action of osmolality on adenosins 3‘,5‘ – monophosphate accumulation in rat renal inner medulla: evidence for relationship to calcium-responsive arachidonate release and prostaglandin synthesis. J Clin Invest 65: 529-542.
42. Stebbins CL, Carretero OA, Mindroiu T, Longhurst JC, 1990 Bradykinin release from contracting skeletal muscle of the cat. J Appl Physiol 69: 1225-1230.
43. Leaf DA, Macrae HS, Grant E, Kraut J, 2003 Isometric exercise increases the size of forearm veins in patients with chronic renal failure. Am J Med Sci 325: 115-119.
44. Rus R, Ponikvar R, Κenda RB, Buturovic-Ponikvar J, 2005 Effects of handgrip training and intermittent compression of upper arm veins on forearm vessels in patients with end-stage renal failure. Ther Apher Dial 9: 241-244.
45. Mustata S, Chan C, Lai V, Miller J, 2004 Impact of an exercise program on arterial stiffness and insulin resistance in hemodialysis patients. J Am Soc Nephrol 15: 2713-2718.
46. Parsons TL, Toffelmire EB, King-VanVlack CE, 2006 Exercise training during hemodialysis improves dialysis efficacy and physical performance. Arch Phys Med Rehabil 87: 680-687.
47. Kong Ch, Tattersall JE, Greenwood RN, Farrington K, 1999 The effect of exercise during haemodialysis on solute removal. Nephrol Dial Transplant 14:2927-2931.
48. Parsons TL, Toffelmire EB, King-VanVlack CE, 2004 The effect of an exercise program during hemodialysis on dialysis efficacy, blood pressure, and quality of life in end-stage renal disease patients. Clin Nephrol 61: 261-274.
Address for correspondence:
Ioannis Karamouzis, 14 Iassonos Zirganou Str., Thessaloniki, Greece, Fax: +30 2310 281234,
E-mail: mkaram@med.auth.gr & karamouzisgiannis@yahoo.gr
Received 10-06-08, Revised 25-01-09, Accepted 25-02-09