Abstract
Objective: To evaluate the efficacy of Laparoscopic Sleeve Gastrectomy (LSG) as a definitive procedure for morbidly obese patients. Design: This constitutes a prospective study carried out in a tertiary care private hospital and included 15 morbidly obese patients who underwent LSG. The operation was performed through two 12 mm and two 5 mm ports, using the Endo-GIA stapler to create a lesser curve gastric tube over a 36-Fr bougie. Results: Operative time, complication rates, hospital length of stay, Body Mass Index (BMI), % of Excess Weight Loss (EWL) and appetite were evaluated. There were six females and nine males, aged (mean±SD) 40.5±10.5 yrs and preoperative BMI 47.8±7.5 kg/m². The operative time was 147.7±43.2 min. There was one conversion to open surgery and one gastric leak with haemorrhage that led to gastric tube stenosis, ultimately requiring revision surgery. All patients, except these two, were discharged on the 2nd postoperative day after an upper GI series and the initiation of a clear liquid diet. At the follow-up (7.5±4.4 months post operatively), the % EWL was 35.7±10.1. Eight patients who received regular postoperative dietician counselling at follow-up did better than the others who did not (% EWL 40.4±3.8 vs 30.2± 4.1, respectively). All patients reported significant loss of appetite.Conclusions: Although the number of patients is relatively small, the data of this study indicate that laparoscopic sleeve gastrectomy is effective in weight reduction, being an acceptable surgical option for morbidly obese patients. A higher number of patients and longer follow-up period will be necessary to evaluate long-term efficacy.
Bariatric surgery remains the only effective treatment for inducing and maintaining satisfactory weight loss and reducing weight-related comorbidities for the morbidly obese patient.1 Bariatric operations have shown an exponential rise both in frequency and diversity during the last few decades paralleling the increasing prevalence of morbid obesity in Western populations.2-4 During this evolutionary process some operations have been abandoned, whereas others have gained wider acceptance.5-8
Laparoscopic Sleeve Gastrectomy (LSG) has been recently introduced in the field9,10 either as the sole bariatric operation for high-risk very obese patients 11 or as the first step followed by Roux-Y gastric bypass or duodenal switch in morbidly obese patients.
Our study focuses on the results of LSG in a prospective series of 15 consecutive patients.
PATIENTS AND METHODS
Six females and nine males, aged (mean±Standard Deviation, SD) 40.5±10.5 yrs, who met the 1991 National Institutes of Health Consensus Development Criteria1 for bariatric operation, were offered laparoscopic sleeve gastrectomy. The mean±SD Body Mass Index (BMI) value of the series was 47.8±7.5 kg/m². All patients had failed medical therapy for their obesity.
Preoperatively the patients were assessed for their general condition and mental status, comorbidities, cardiovascular risk factors and motivation for surgery. Efforts previously made by the patients to lose weight had failed. However, we did not take any specific measures for preoperative weight reduction. A consent form was routinely obtained.
Surgical technique: With the patient intubated in supine position, preumoperitoneum is established through the umbilicus using the open (Hassan) technique. Trocar placement is as follows:
- One 5 mm trocar is placed up to the costochondral junction for the insertion of the liver retractor;
- Two 12 mm trocars are placed on the right and left middle clavicular lines, respectively, accommodating the instruments;
- One 5 mm trocar is placed on the left anterior axillary line.
After the identification of the gastric antrum (measurement of the distal 7 cm from the pylorus on the greater curvature), ultrasonic scissors are used to dissect the greater omentum from the greater curvature of the stomach dividing the gastrocolic and gastrosplenic ligaments up to the esophagogastric junction.
After this critical stage, the first 45 mm – 3.5 mm linear stapler is placed at the point of the initial dissection on the greater curvature, creating a vertical cut on the gastric wall. Then a 36-Fr bougie is inserted and sequential firings of the endoGastro-Intestinal-Anastomosis (GIA) with 60 mm – 3.5 mm linear staplers are applied over it, up to the esophagogastric junction (Figure 1). The gastric remnant is then easily extracted through the umbilicus. No drains are needed and a nasogastric tube is left in place.
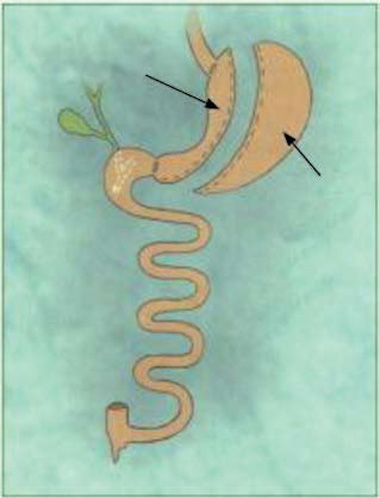
Figure 1. Schematic representation of sleeve gastrectomy.
Postoperative care: A standard clinical protocol was used for all patients. The nasogastric tube was removed on the first postoperative day after a normal upper GI series with gastrographin (Figure 2). Thirteen patients felt able to return home and were discharged on the 2nd postoperative day with instructions to follow a liquid diet for four weeks.
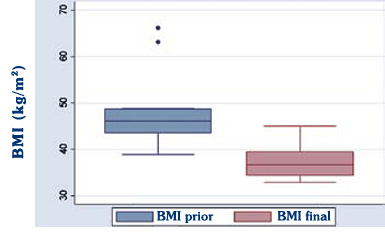
Figure 2. Boxplots of Body Mass Index (BMI) prior to surgery (BMI prior) and at last examination (BMI final) at 7.5±4.4 (mean±SD) months after the operation.
A clinical dietician followed up eight patients in an orderly fashion: outpatient visits were scheduled once a month for the first three postoperative months and every three months thereafter. Seven patients were followed up via regular phone communication by a research nurse.
Baseline measures: Demographic data were collected at the preoperative evaluation. Perioperative outcome measures including operative time, hospital stay, intraoperative and postoperative complications.
Follow-up measures: Data on BMI, total weight loss, % Excess Weight Loss (EWL) and perception of appetite were evaluated at 1, 3, 6 and 12 months postoperatively.
Statistical Methods:Categorical data were summarized as frequencies or percentages (%). Continuous data were summarized as mean ±SD. The Kolmogorov-Smirnov test was used to evaluate normal distribution, which means that p-values >0.05 indicate normally distributed variables. Comparisons of BMI before and after gastrectomy were evaluated using the paired samples t test. BMI before and after gastrectomy were normally distributed according to the Kolmogorov-Smirnov test for normality. A p-value of 0.05 was considered statistically significant. Data were analyzed using STATA 9.1 and SAS statistical software.
RESULTS
Between November 2005 and November 2007, six females and nine males, aged 40.5±10.5 yrs, underwent laparoscopic sleeve gastrectomy. Patients body weight (Table 1 ) (mean±SD) was 138.8±26.5 kg and BMI 47.8±7.5 kg/m² preoperatively. Operative time was 147.6±43.2 min.
Postoperatively, at a mean follow-up of 7.4±4.4 months, weight loss was 32.8±3.3 kg, the BMI was 37.0±11.0 and the %EWL was 35.7±10.1 (Table 1, Figure 2).
We had one conversion to open surgery (8.3%) due to full thickness tear of the gastric wall (accidental grasping of the pliable, soft terminal portion of the bougie along with the gastric wall).
Another patient exhibited a tiny gastric leak with staple line haemorrhage on the first postoperative day. In this particular case, we had used biosynthetic absorbable material to reinforce the staple line that we believe negatively affected the closure and led to hemorrhage. The patient was urgently reoperated on due to hemoperitoneum, but severe gastric tube stenosis eventually mandated revision surgery after about one month postoperatively.
A small left subdiaphragmatic haematoma was found in one patient that required no transfusion therapy and was treated on an outpatient basis without any other untoward events. No mortality was encountered.
Except for the converted case, the patient with the leak at the staple line and the acute haemorrhage, all other patients felt able to return home and were discharged on the 2nd postoperative day without evidence of leakage during an upper GI series carried out on the previous day (Figure 3).
Figure 3. First postoperative day upper gastrointestinal imaging.
BMI postoperatively was significantly reduced to 37.1±3.3 kg/m² from a preoperative value of 47.8±7.5 (P<0.001).
Schematic representation of BMI reduction over time is shown in Figure 4.
Eight patients who received regular postoperative dietician counselling at follow-up did better than the others who did not (%EWL 40.4±3.8 vs 30.2±4.1).
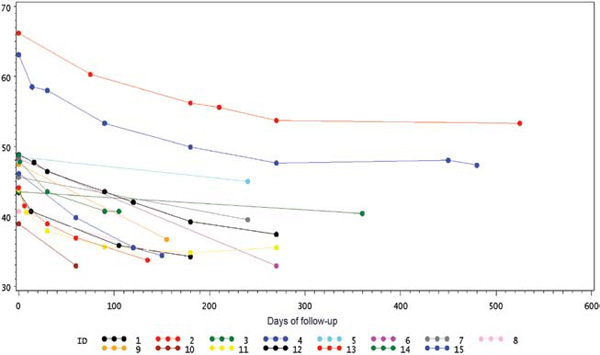
Figure 4. Body Mass Index (BMI) values in each patient at follow-up.
DISCUSSION
LSG as a definitive bariatric procedure was quite successful for body weight reduction in our series. Following the operation, a statistically significant mean weight loss of 32.7 kg over a mean period of 7.4 months leading to a mean %EWL of 35.7 was observed. Other authors have disclosed analogous results.
In one of the first studies on the efficacy of LSG from Gagner et al,9 20 patients who underwent LSG were compared with 57 patients who had undergone Intragastric Balloon. The study showed that at six months patients undergoing LSG showed a faster and greater weight loss than those using a intragastric ballon. Moreover, LSG was found to be a safe procedure, with reproducible results, in contrast to the intragatric ballon which was tolerated by 93% of the patients. The results of our study indicate that both mean weight loss and %EWL were significant following LSG. In a study from Mognol P et al10 which included ten patients, the average %EWL and BMI reduction at one year were 51% and 23%, respectively. It was concluded that LSG can be safely integrated into a bariatric surgical program with good results in terms of weight loss and quality of life.
In a study conducted by Cottam D et al11 involving 126 patients with a preoperative mean BMI value of 65.3±0.8, the mean %EWL after LSG at one year was 46. One distant mortality was encountered, and the incidence of major complications reached 13%. Rosenthal RJ et al12 reported 30 patients with a mean preoperative BMI value of 41.4. Mean weight loss in this study at three and six months postoperatively was 22.7 kg and 30.5 kg and mean % EWL 40.7 and 52.8, respectively.
These figures along with ours compare favourably with those reported for adjustable Gastric Banding13-15 and even for Roux-en-Y Gastric Bypass,15-17 series thus far reported. In a recent prospective randomized trial of LSG vs gastric banding (GB),13 the median %EWL at one year was 41.4 after GB and 57.7 after LSG (P-value=0.001) and at three years 48 after GB and 66 after LSG (P-value=0.003). Moreover, weight loss and loss of feeling of hunger after one year and three years were better after SG than GB.
In our patients, weight loss was found to be steady in all patients throughout the follow-up. A consistent feature of our small series is the report of significant loss of feeling of hunger by all patients. Permanent reduction of plasma ghrelin levels may play a role in this phenomenon as stated by various reports.18,19 Ghrelin levels reduction, as an inherent sequel of sleeve gastrectomy associated reduction of gastric fundus mass, constitutes a significant, hormone mediated advantage, over the other bariatric procedures.
Technically, this operation appears to be less demanding than the Roux-en-Y gastric bypass. However, certain specific operative points must be emphasized. First of all the use of GIA staplers has been proven safe for the majority of cases. In addition, we have found no advantage to using specialized bioabsorbable staple reinforcement materials. Although difficult to prove, it is hypothesized that the one gastric leak in the present series could be attributed to malfunction of the stapler covered with the reinforcement material.
A second issue involves the bougie that is used to create the gastric tube. We have found it difficult to stabilize it across the lesser curve, indicating that care should be taken regarding possible accidental grasping by the GIA stapler.
Onset and duration of prophylactic anticoagulation constitutes another challenging issue. Since the operation involves the creation of a lengthy suture line that is especially prone to hemorrhage, sound clinical judgment should be exercised. We rely on intraoperative pneumatic compression stockings devices and early mobilization of the patient. Low-molecular weight heparin is started on the 1st postoperative day when the patient is hemodynamically stable and blood hemoglobin shows no serious decrease. We had no pulmonary embolism episodes but the hemorrhagic event and the hematomas arouse some skepticism as to the anticoagulation scheme.
Nevertheless, LSG has been associated with several important advantages in terms of pylorus preservation (avoidance of dumping syndrome) and preservation of intestinal continuity (no intestinal bypass and malabsorption). It also required 2.26 days mean hospital stay and no reliance on future adjustments thus far.
A significant finding of the present study was that a group of eight patients that were followed up and received regular postoperative dietician counselling did remarkably better than the others who did not (mean %EWL 40.4 vs 30.2. This underlines the need for a ‘team approach’ in the field of bariatric surgery
REFERENCES
1. Gastrointestinal surgery for severe obesity: Proceedings of a National Institutes of Health Consesus Development Conference, 1992 Am J Clin Nutr 55: Suppl 2: 487-619.
2. Kluthe R, Schubert A, 1985 Obesity in Europe. Ann Intern Med 103: 1037-1042.
3. Macdonald SM, Reeder BA, Chen Y, Després JP, 1996 Obesity in Canada: a descriptive analysis. Canadian Heart Health Surveys Research Group. Can Med Assoc J 157: Suppl 1: 3-9.
4. Flegal KM, Carroll MD, Kuczmarski RJ, Johnson CL, 1998 Overweight and obesity in the United States: prevalence and trends, 1960-1994. Int J Obes Relat Metab Disord 22: 39-47.
5. McFarland RJ, Gazet JC, Pilkington TR, 1985 A 13-year review of jejunoileal bypass. Br J Surg 72: 81-87.
6. Mason EE, Maher JW, Doherty C, et al, 1992 Ten years of vertical banded gastroplasty for severe obesity. Probl Gen Surg 9: 280-289.
7. Linner JH, 1984 Gastric operations. Linner JH (ed), Surgery for Morbid Obesity. New York, Springer, p, 65-107.
8. Santry HD, Gillen DL, Lauderdale DS, 2005 Trends in bariatric surgical procedures JAMA 294: 1909-1916.
9. Milone L, Strong V, Gagner M, 2005 Laparoscopic sleeve gastrectomy is superior to endoscopic intragastric balloon as a first stage procedure for super-obese patients (BMI > or =50) Obes Surg 15: 612-617.
10. Mognol P, Chosidow D, Marmuse JP, 2005 Laparoscopic sleeve gastrectomy as an initial bariatric operation for high-risk patients: initial results in 10 patients. Obes Surg 15: 1030-1033.
11. Cottam D, Qureshi FD, Mattar SG, et al, 2006 Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity Surg Endosc 20: 859-863.
12. Roa PE, Kaidar-Person O, Pinto D, Cho M, Szomstein S, Rosenthal RJ, 2006 Laparoscopic sleeve gastrectomy as treatment for morbid obesity: technique and short-term outcome. Obes Surg 16: 1323-1326.
13. Himpens J, Dapri G, Cadière GB, et al, 2006 A prospective randomized study between laparoscopic gastric banding and laparoscopic isolated sleeve gastrectomy: results after 1 and 3 years. Obes Surg 16: 1450-1456.
14. Martin LF, Smits GJ, Greenstein RJ, 2007 Treating morbid obesity with laparoscopic adjustable gastric banding. Am J Surg 194: 333-343.
15. Favretti F, Segato G, Ashton D, et al, 2007 Laparoscopic adjustable gastric banding in 1,791 consecutive obese patients: 12-year results. Obes Surg 17: 168-175.
16. O’Brien PE, McPhail T, Chaston TB, Dixon JB, 2006 Systematic review of medium-term weight loss after bariatric operations. Obes Surg 16:1032-1040
17. Christou NV, Look D, Maclean LD, 2006 Weight gain after short-and long-limb gastric bypass in patients followed longer than 10 years. Ann Surg 244: 734-740.
18. Langer FB, Reza Hoda MA, Bohdjalian A, et al, 2005 Sleeve gastrectomy and gastric banding: effects on plasma ghrelin levels. Obes Surg 15: 1501-1502.
19. Kotidis EV, Koliakos GG, Baltzopoulos VG, Ioannidis KN, Yovos JG, Papavramidis ST, 2006 Serum ghrelin, leptin and adiponectin levels before and after weight loss: comparison of three methods of treatment-a prospective study. Obes Surg 16: 1425-1432.
Address for correspondence:
Linos D., Harvard Medical School, Director of 1st Surgical
Clinic, “Hygeia” Hospital, 7 Fragoklisias Street,
151 25 Marousi, Athens, Greece, Tel.: +30 210 6125001,
Fax: +30 210 6126170, E-mail: dlinos@hms.harvard.edu
Received 27-06-08, Revised 20-01-09, Accepted 25-02-09