Address Correspondence to:
44, Tzanakaki Str., 73100 Chania (Crete), Greece, Tel.: +30821052052, fax: 30821052752, e-mail: manvar@cha.forthnet.gr, Athens Naval and Veterans Hospital, 2nd Surgical Department, Athens, Greece, Tel. / fax: +30106443854
Received 10-09-2001, Revised 13-11-2001, Accepted 18-10-2001
Abstract
The purpose of this study was to investigate the value of “high-resolution ultrasonography” in the differential diagnosis of benign from malignant thyroid nodules. The study included ultrasonography examination of 77 patients with thyroid nodules, who underwent thyroidectomy. The histological findings were subsequently compared with pre-operative sonographic findings. Thirty patients had malignant nodules and 47 patients had benign nodules. The sonographic features that were
studied were: hypoechogenicity or isoechogenicity of the nodules, the presence of peripheral halo, the regular or irregular contour, the presence of microcalcifications, the presence of cystic degeneration and the existence of solitary or multiple nodules. The results revealed that low echogenicity constitutes a sonographic feature of malignancy with high sensitivity and medium specificity. The presence of microcalcifications is a feature of malignancy with low sensitivity but high specificity. Conversely, the regular contour of a nodule and the presence of peripheral halo are features of benign nodule with high sensitivity and specificity. Cystic degeneration occurs at the same rate in benign and malignant nodules. Solitary nodules were malignant more often than were multiple nodules, a finding that was not, however, statistically significant. We conclude that high-resolution ultrasonography contributes greatly in the preoperative differential diagnosis of thyroid nodules. It constitutes an important diagnostic means complementary to needle biopsy in the management of thyroid nodules and it is even more valuable in those nodules that cannot undergo biopsy.
INTRODUCTION
Thyroid nodules are very frequent in the general population. In the USA, their frequency is reported to vary from 4% to 21%1,2. The frequency of diagnosed nodules increases with age and it is approximately 4 times higher in women than in men. Recently, the frequency of diagnosed nodules has increased due to the widespread use of ultrasonography. Thus sonographically diagnosed nodules by various researchers vary from 21% to 67% in persons aged over 503-5. On the other hand, thyroid carcinoma is rare since it constitutes 1% of all carcinomas encountered in humans and the possibility that a thyroid nodule may harbour a carcinoma is relatively small approximating 5%6. Moreover, most thyroid carcinomas are papillary carcinomas which develop slowly and have a generally good prognosis7.
Therefore, the dilemma for the physician is how to diagnose as many malignant nodules as possible without, subjecting many benign nodules to unnecessary surgery. Pre-operative differential diagnosis of benign from malignant thyroid nodules is a relatively strenuous process because there is no pre-operative method with 100% sensitivity and specificity in the diagnosis of thyroid cancer. The pre-operative examinations usually performed are biochemical examinations such as the assessment of TSH, calcitonin and thyroid autoantibodies which, however, offer little help in the diagnosis of the common types of thyroid cancer. Furthermore, the isotopic scintiscan which differentiates cold from hot nodules has a high sensitivity but low specificity since almost all malignant nodules are cold but out of the cold nodules only 15-20% are malignant8. The fine needle aspiration biopsy has a higher sensitivity (85-95%) and specificity (85-95%) than any other method and is nowadays the most important preoperative examination in the differential diagnosis of thyroid nodules9,10. However, it is an invasive method to which the patient does not always consent and cannot be applied to non-palpable nodules and to nodules located on the rear surface of the gland. Ultrasonography provides information on the size and the location of a nodule and is very useful in the correct interpretation of the scintigraphy. Moreover, it offers valuable help in performing ultrasound guided needle aspiration biopsy on relatively small non-palpable nodules11.
It seems that the new generation high-resolution sonographers of superficial organs make it possible to acquire useful information in differentiating benign from malignant thyroid nodules12,13. High frequency transducers (10-13 MHz) have high axial and lateral resolution and are capable of distinguishing small sonographic changes of the thyroid parenchyma such as microcalcifications, thin peripheral halo, cystic degenerations and small differences in echogenicity between the nodule and the healthy parenchyma.
The purpose of this study was to review the sonographic findings of thyroid nodules, which were examined with high-resolution equipment at our practice and which were subsequently excised in order to assess the value of high-resolution ultrasonography in the differential diagnosis of benign from malignant nodules.
MATERIALS – METHODS
During the last decade, we examined about 10,000 patients sonographically at our practice. Approximately 1,000 patients had thyroid nodules of different size. Four hundred of these patients had thyroid nodules larger than 1 cm (maximal diameter) and underwent thyroidectomy. In the histological examination, 30 patients proved to have malignant nodules and the remaining 370 patients had benign nodules. In our study we included all 30 patients with malignant nodules. From the total 370 patients with benign ones we randomly chose 47. To ensure that the sample was random, we chose 1 out of every 8 patients, based on their medical record number. The histological examination showed that out of the 30 malignant nodules, 21 constituted papillary carcinomas, 5 follicular carcinomas, 2 Hurthle cell carcinomas, 1 anaplastic carcinoma and 1 lymphoma. Out of the 47 benign nodules, 45 were hyperplastic nodules and 2 were follicular adenomas. Of the patients who had the malignant nodules, 25 were women and 5 men with a mean age of 44.5 (+13.7) whereas of the patients with the benign nodules, 41 were women and 6 men with a mean age of 49 (+13.7). The scintigraphic examination showed all malignant nodules to be cold, whereas of the benign nodules 36 were cold and 11 were hot.
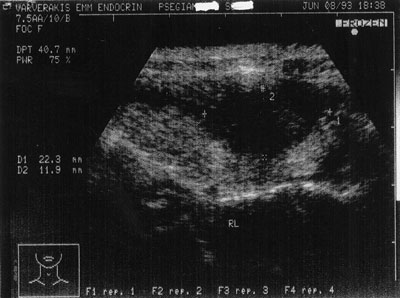
Figure 1. Hypoechogenic nodule, 22×11 mm. in size with irregular contour. It was a papillary carcinoma.
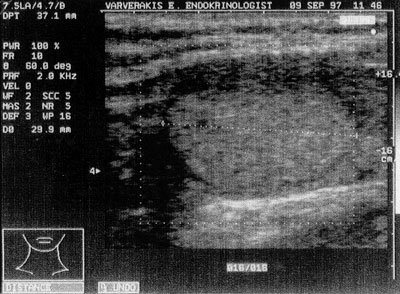
Figure 2. Isoechogenic nodule, 29 mm. in size with peripheral halo. It was a hyperplastic nodule.
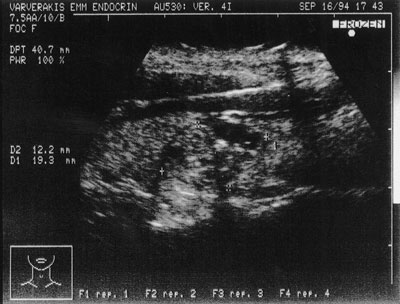
Figure 3. Hypoechogenic nodule, 12×19 mm. in size with microcalcifications. It was a papillary carcinoma
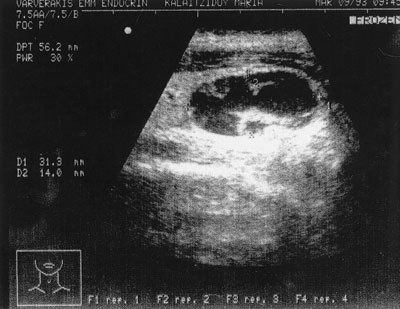
Figure 4. Cystic nodule, 31×14 mm. in size with solid elements on the wall. It was a hyperplastic nodule
All ultrasonographic examinations were carried out by the same operator (E.V.) using the same sonographer (ESAOTE – BIOSOUND, USA, Model AU4). This sonographer has 2 transducers, one that operates at 7.5-10 MHz frequencies and another that operates at 10-13 MHz frequencies. The 7.5-10 MHz transducer has an axial resolution capacity of 0.2 mm and max focal depth of 6 cm, whereas the 10-13 MHz transducer has an axial resolution capacity of 0.1 mm and max focal depth of 5 cm. The majority of the patients were examined with the 10-13 MHz transducer, apart from few patients with deeply localized nodules who were examined with the 7.5-10 MHz transducer. Postoperatively, the histological findings were compared with the preoperative sonographic findings.
The sonographic characteristics studied were the hypoechogenicity (Figure 1) or isoechogenicity (Figure 2) of the nodules in relation to the normal thyroid parenchyma; the presence of peripheral halo (Figure 2); the presence of a regular (Figure 2) or irregular (Figure 1) contour of the nodule; the presence of microcalcifications (<2mm) in the nodule, (Figure 3); the presence of cystic degeneration, which appears as an unechoic area inside the nodule (Figure 4); the presence of solitary or multiple nodules inside the thyroid gland.
Statistical analysis of the findings was carried out using the X2 test with Yates correction. A p-value of less than 0.05 was considered to indicate statistical significance.
RESULTS
The results are shown in figure 5.
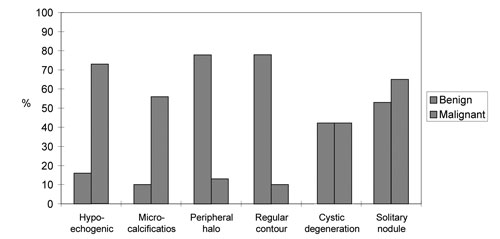
Figure 5. Frequencies of sonographic characteristics in benign and malignant nodules.
Twenty two out of 30 (73%) malignant nodules and 16 out of 47 (34%) benign ones were hypoechogenic. This difference was statistically significant (p<0.01), sensitivity: 22/30=0.73, specificity: 31/47=0.65, positive predictive value 9 (p.p.v.): 22/(22+16)=0.57.
A peripheral halo appeared in 37 out of 47 (78%) benign nodules and in 4 out of 30 (13%) malignant ones, p<0.001 sensitivity: 37/47=0.78, specificity: 26/30=0.86, p.p.v: 37/41=0.90.
Thirty seven out of 47 (78%) benign nodules and 3 out of 30 (10%) malignant ones had a regular contour, p<0.001, sensitivity: 37/47=0.78, specificity: 27/30=0.90, p.p.v: 37/40=0.92.
Seventeen out of 30 (56%) malignant nodules and 5 out of 47 (10%) benign ones had microcalcifications, p<0.001, sensitivity: 17/30=0.56, specificity: 42/47=0.89, p.p.v.: 17/22=0.77. It is worth mentioning that 16 out of 21 (76%) papillary carcinomas had microcalcifications.
Malignant nodules tended to be solitary more frequently than benign ones (20/30, 65% vs. 25/47, 53%); however, this difference was not statistically significant, 0.01<0.5.
There was no difference in the incidence of cystic degeneration between malignant (43%) and benign (44%) nodules, (p>0.5).
DISCUSSION
There is a significant amount of disagreement among endocrinologists as far as thyroid nodule management is concerned. Specialists in the field were asked to express their opinion about thyroid nodule management in two reviews: a North-American review14, and a European one15. According to the results, North-American endocrinologists prefer FNA at 100%, ultrasonography at 34% and scintigraphy at 34%, whereas European specialists prefer FNA at 99%, ultrasonography at 80% and scintigraphy at 66%. Therefore, it appears that apart from FNA, which constitutes a major examination preferred and used by everyone at the same frequency, there is a significant difference in the use of imaging examinations, particularly sonography, in the management of thyroid nodules. This is probably because old technology sono-graphers provided information concerning only the size and location of the nodules and did not contribute in distinguishing benign from malignant ones. The definition of criteria of malignancy using high-resolution sono-graphers, such as the one used in our study, is, therefore, very important.
In the nodules that were examined in our study, hypoechogenicity was characteristic of malignancy with high sensitivity (73%) but lower specificity (65%). Most malignant nodules are, therefore, hypoechoic but hypoechogenicity does not necessarily mean that the nodule is malignant. Other researchers have reported analogous results. Naoun et al16 observed that 92% of thyroid carcinomas were hypoechoic, whereas 18% of benign nodules were hypoechoic. Lin et al17 showed that 76% of thyroid carcinomas were hypoechoic, whereas 23% of benign nodules were hypoechoic. It is worth mentioning that hypoechogenicity is also characteristic of Hashimoto’s thyroiditis where, however, the whole of the thyroid parenchyma is usually involved18. A hypo-echogenic nodule may thus be difficult to evaluate against a background of autoimmune thyroiditis. In these cases, it is necessary to investigate other sonographic features such as the peripheral halo, microcalcifications, or the presence of lymph nodes.
In our study, a regular contour was characteristic of a benign nodule with high sensitivity (78%) and very high specificity (90%). Other researchers have come to similar conclusions19. A regular contour may be combined with low echogenicity, as in a pure cyst and in colloid nodules or with isoechogenicity, as in the glandular nodules. On the other hand, an irregular contour is characteristic of malignancy with high sensitivity and specificity
Peripheral halo, in our study, was characteristic of a benign nodule with high sensitivity (78%) and very high specificity (86%). It is a hypoechoic halo in the circumference of the nodule. The colour flow Doppler sono-graphy shows that it is due to vessels arranged peripherally to the nodule. The peripheral halo of benign nodules is thin (<2 mm), regular and usually surrounds the nodule without splitting. Sometimes halo is also found in malignant nodules. In this case however the halo is thicker and irregular20.
Microcalcifications are shown on the ultrasonography as hyperechoic dots inside the nodule. They are less than 2 mm in size and are usually multiple and concentrated. Sometimes acoustic shadow is shown behind them. In our study, microcalcifications were characteristic of malignancy with low sensitivity (56%) but very high specificity (89%). Their presence, therefore, is an almost pathognomic characteristic of thyroid carcinoma, and in most cases a papillary one. According to Amodio et al21, microcalcifications have a greater specificity for malignancy than all other sonographic features. The sensitivity of this finding, in particular, is higher with a high frequency transducer (13 MHz) than with a lower frequency one (7,5 MHz). Kakkos et al22 also reported that the highest frequency of calcifications is found in the thyroid carcinoma (54%) followed by the multinodular goiter (40%), the solitary glandular nodule (14%) and the follicular adenoma (12%). At this point, it is worth mentioning that calcifications are also found in Hashimoto thyroiditis and in multinodular goiter. Moreover, it is a very common finding on those parts of the thyroid gland that underwent needle aspiration. It is, therefore, very important to evaluate calcifications in relation to their morphology, the patient’s medical history, the clinical profile and the biochemical data (TSH, thyroid autoantibodies). Large calcifications (>2mm) observed on different parts of a multinodular goiter, as well as peripheral, eggshell-like, calcifications are relatively common in benign nodules. On the other hand, small (<2mm) calcifications inside a hypoechoic nodule are characteristic of malignancy. According to Solbiati20, while peripheral (eggshell) calcification is characteristic of a benign nodule, microcalcifications within the nodule are characteristic of malignancy with low sensitivity (59%) but high specificity (95%).
In our study, cystic degeneration had the same frequency (43%) in benign as well as in malignant nodules. In a retrospective study on 153 operated cystic nodules, Hiromura23 reported that cancer frequency in cystic thyroid nodules varies from 4 to 80%, depending on the morphology of the nodules. In purely cystic nodules it is only 4%, whereas in cystic nodules with solid elements on the wall, its frequency amounts to 80%. In our study, all nodules had solid together with cystic elements and none of them was purely cystic. Moreover, the 2 nodules that had the morphology of a cyst with solid elements on the wall were both benign. Furthermore, Watters et al24 reported that malignant nodules at ultrasonography examination tend to be solid and hypoechoic without halo, with cystic degeneration at 26% and microcalcifications at 37%.
In our study, solitary nodules tended to be malignant more often than multiple ones, a finding that was not, however, statistically significant. Other researchers have also come to the same conclusion25. On the other hand, older studies which were mostly based on palpation, during which small nodules were not felt, showed that thyroid cancer is mostly found in the form of a solitary nodule. It seems, however, that a palpable solitary nodule represents a sonographically multinodular gland in about 50% of patients26.
We conclude that preoperative examination of thyroid noduls with a high-resolution sonographer not only provides useful information on the size and location of thyroid nodules, but also contributes significantly in the differential diagnosis of benign from malignant nodules. As it is a simple, inexpensive and radiation-free examination with high sensitivity and specificity, we believe that high-resolution ultrasonography can be a valuable means in the preoperative diagnosis of thyroid nodules, complementary to fine needle aspiration. More specifically, it is of great value in the case of nodules that cannot undergo needle biopsy. However, in order to be able to obtain all necessary information, it is important that a high-resolution sonographer of superficial organs is used and the operator has the necessary experience.
REFERENCES
1. Mazzaferi El, de los Santos ET, Rofagha-Keyhani S, 1988 Solitary thyroid nodule: Diagnosis and management. Med Clin North America 72(5): 1177-211.
2. Gharib H, 1997 Changing concepts in the diagnosis and management of thyroid nodules: Endocrinol and Metab Clin North Am 26(4): 777-800.
3. Ezzat S, Sarti DA, Cain DR, Braunstein GD, 1999 Thyroid incidentalomas. Prevalence by palpation and ultrasonography: Arch Int Med; 22 154(16): 1838-40.
4. McCaffrey TV, 2000 Evaluation of the thyroid nodule: Cancer control 7(3): 223-8.
5. Miki H, Oshimo K, Inoue H, Kawano M, Tanaka K, Komaki K, Uyama T, Morimoto T, Monden Y, 1993 Incidence of ultrasonographically-detected thyroid nodules in healthy adults: Tokushima J Exp Med 40(1-2): 43-6.
6. Castro MR,Gharib H, 2000 Jan Thyroid nodules and cancer. When to wait and watch, when to refer. Postgrad Med 107(1): 113-6, 119-20, 123-4.
7. Gerard N Burrow, The Thyroid: Nodules and Neoplasia. In: Felig P, Baxter J, Broadus A, Frohman L (eds) Endocrinology and Metabolism 2nd edition: Mc Graw-Hill Book Company: Inc, pp 473-504.
8. Danese D, Sciacchitano S, 1993 The thyroid nodule. Diagnostic considerations: Minerva Endocrinol 18(3): 129-37.
9. Amrikachi M, Ramzy I, Rubenfeld S, Wheeler TM, 2001 Accuracy of fine needle aspiration of thyroid: Arch Pathol Lab Med 125(4): 484-8.
10. Tabaqchali MA, Hanson JM, Johnson SJ, Wadehra V, Lennard TW, Proud G, 2000 Thyroid aspiration cytology in Newcastle: a six-year cytology/histology correlation study: Ann R Coll Surg Engl 82(3): 149-55.
11. Raush P, Nowels K, Jeffrey RB, 2001 Ultrasonographically guided thyroid biopsy: a review with emphasis on technique: J Ultrsound Med 20(1): 79-85.
12. Haber RS, 2000 Role of ultrasonography in the diagnosis and management of thyroid cancer: Endocr Pract 6(5): 396-400.
13. Leenhard L, 1993 Comments on the ultrasonographic evaluation of thyroid nodules: Ann Endocrinol (Paris) 54(4): 237-40.
14. Bennedbaek FN, Hegedus L, 2000 Management of the solitary thyroid nodule: results of a North American survey: J Clin Endocrinol Metab 85(7): 2493-8.
15. Bennedbaek FN, Perrild H, Hegedus L, 1999 Diagnosis and treatment of solitary thyroid nodule. Results of a European survey: Clin Endocrinol (Oxf) 50(3): 357-63.
16. Naoun A, 1993 Evaluation of ultrasonography in the diagnosis of thyroid nodules. Ann Endocrinol (Paris) 54(4): 232-4.
17. Lin JD, Hsuen C, Chao TC, Weng HF, Huang BY, 1997 Thyroid follicular neoplasms diagnosed by high-resolution ultrasonography with fine needle aspiration cytology: Acta Cytol 41(3): 687-91.
18. Espinasse P, Desuzines C, Cercasi JC, 1984 Measurements of an index of echogenity in some thyroid diseases: J Clin Ultrasound 12(1): 33-6
19. Koike E, Nogguchi S, Yamashita H, Murakami T, Ohsihima A, Kawamoto H, 2001 Ultrasonographic characteristics of thyroid nodules: prediction of malignancy: Arch Surg 136(3): 334-7.
20. Solbiati L, Cioffi V, Ballarati E, 1992 Ultrasonography of the neck: Radiologic Clinics of North America Vol 30(5): 941-954.
21. Amodio F, Carbone M, Rossi E, Brunese L, Pisano G, Iorio S, Benincasa G, Vallone G, 1999 An update of B-mode echography in the characterisation of nodular thyroid diseases. An echographic study comparing 7.5 and 13 MHz probes: Radiol Med (Torino) 98(3): 178-82.
22. Kakkos SK, Skopa CD, Chalmoukis AK, Karachalios Da, Spiliotis JD, Harkoftakis JG, Karavias DD, Androulakis JA, Vagenakis AG, 2000 Relative risk of cancer in sonographically detected thyroid nodules with calcifications: J Clin Ultrasound 28(7): 347-52.
23. Hiromura T, 1994 Ultrasonography of cystic thyroid nodules: sonographic-pathologic correlation: Nippon-Igacu-Hoshasen-Gakkai-Zasshi 25; 54(6): 500-9.
24. Watters DA, Ahuja AT, Evans RM, Chick W, King WW, Metreweli C, Li AK, 1992 Role of ultrasound in the management of thyroid nodules: Am J Surg 164(6): 654-7.
25. Sachmechi I, Miller E, Varatharajah R, Chernys A, Carrol Z, Kissin E, Rosner F, 2000 Thyroid carcinoma in single cold nodules and in cold nodules of multinodular goiters: Endocr Pract 6(1): 110-2
26. Tan GH, Gharib H, Reading CC, 1995 Solitary thyroid nodule. Comparison between palpation and ultrasonography: Arch Intern Med 155(22): 2418-23.