Abstract
OBJECTIVE: Glucocorticoids (GC) are the treatment of choice for moderate-to-severe and active Graves’ orbitopathy (GO), but optimal treatment is still . The aim of the present study was to analyze the efficacy and tolerability of combined parenteral GC pulse therapy followed by oral GC in the interpulse period.
DESIGN: The study included 50 patients (48±10 years; 37 female) with untreated, active and moderate-to-severe GO. Patients received 500mg of methylprednisolone in 500ml of physiologic saline. Infusion was repeated after 48h and then followed by tapering doses of oral prednisone and the cycle repeated each month for the next 5 months. The cumulative dose was 10.2g.Ophthalmic assessment was performed before and 6 months after start of treatment. Side effects of GC therapy were evaluated and recorded each month.
RESULTS: GC showed the greatest effectiveness on soft tissue changes (incorporated in the CAS). Median CAS values decreased from 4.5 to 2 (p>0.001). Improvement was demonstrated in 37 patients (74%), there was no change in 13 patients (26%) and none of the patients presented with deterioration of inflammatory status. Diplopia improved in 21 patients (42%), was unchanged in 28 patients (56%) and deteriorated in 1 patient (2%). Improvement in visual acuity occurred in 36% of patients. At 6 months, 33/50 patients (66%) demonstrated overall treatment response. Response to GC therapy was influenced by CAS, TSHRAb and smoking behavior. The only independent parameter associated with positive treatment response was CAS ≥4 (p<0.001). Side effects occurred in 35/50 patients (70%) and the vast majority of them were mild to moderate. During the 6-months follow-up period, 2/33 patients (6%) had relapsing GO.
CONCLUSION: With appropriate selection of patients and careful monitoring during and after treatment, combined parenteral and oral GC therapy is effective and safe.
INTRODUCTION
Graves’ orbitopathy (GO) is an autoimmune disorder of the retrobulbar tissue1 that is often associated with Graves’ disease, but it may also occur in patients who are hypothyroid due to chronic autoimmune Hashimoto thyroiditis, as well as in patients with normal thyroid function (euthyroid ophthalmopathy).2
Approximately half of the patients with Graves’ disease have ocular involvement, but it is usually mild; clinically relevant disease occurs in 20-30% and is sight-threatening in 3-5% of patients.3 Glucocorticoids (GC) are the first-line treatment for moderate-to-severe and active GO (EUGOGO criteria).4 In the last 20 years, several studies have documented the effectiveness of GC by the oral, intravenous or intrabulbar route,3,5 but the optimal treatment involving no or minimal side effects is still .
Although the intravenous route is considered to be more effective and better tolerated than the oral route, severe adverse events, including fatal cases, are more common in patients who receive intravenous GC, usually exceeding 8g, though it may occur on low doses as well.5,6
The aim of the present study was to analyze the efficacy and tolerability of combined parenetral GC (IVGC) pulse therapy followed by oral GC (ORGC) in the interpulse period.
METHODS
Patients
The study included 50 consecutive patients (48±10 years; 37 female, 13 male) who were admitted to our hospital for GC pulse therapy between April 2004 and December 2006. Inclusion criteria was untreated, active and moderate-to-severe GO, whereas exclusion criteria was contraindication to glucocorticoids. All patients were euthyroid when starting pulse therapy and remained euthyroid during the treatment.
Complete blood biochemistry, clinical evaluation including electrocardiography, chest x- ray, thyroid and abdominal ultrasound were performed before the start of the treatment. Informed consent was obtained from all patients.
GC therapy
Patients received 500mg of methylprednisolone in 500ml of physiologic saline as a 4h intravenous infusion. Infusion was repeated after 48h. On the day after the second infusion patients started with oral prednisone according to the following schedule: 40mg/day for the first week, 30mg/day for the second week, 20mg/day for the third week and 10mg/day for the fourth week.7 This sequence of therapy was repeated each month for the next 5 months. Thus, the cumulative dose was 10.2g. Side effects of GC therapy were evaluated and recorded each month.
Ocular evaluation
Patients were evaluated at baseline and after 6 months. Eye examinations were performed by the same ophthalmologist and included evaluation of: eyelid aperture size (measured in mm), any soft tissue involvement (with reference to the Color Atlas at www.eugogo.eu),8 intraocular pressure, visual acuity and proptosis which was evaluated using the Hertel exophthalmometer.
Eye muscle involvement was assessed using the Gorman diplopia score: no diplopia (absent), diplopia when patient is tired or awakening (intermittent), diplopia at extremes of gaze (inconstant) and continuous diplopia in the primary and reading position (constant).9 Inferior rectus eye muscles thickness was evaluated by ultrasound.
Assessment of GO activity was carried out using the clinical activity score (CAS) which consists of seven items: spontaneous retrobulbar pain, pain during eye movement, conjunctival redness, redness of the eyelids, chemosis, swelling of the caruncle and swelling of the eyelids; the final score was calculated as the sum (1 point for each) of all items.10
The overall response to the therapy after 6 months was defined as a relevant improvement of at least the two major criteria (decrease in proptosis and lid width of 2mm each, changes in degree of diplopia, changes in the CAS of 2 points or more and changes of 1/10 or more in visual acuity) and one minor criterion (soft tissue changes, self assessment evaluation).11
Quality of life (QoL) was evaluated with the validated disease specific GO-QoL questionnaire.12 In addition, all adverse events and biochemistry analysis were recorded at monthly intervals during hospitalization. Patients were followed for the next 6 months.
Evaluation of thyroid function
Thyroid function was assessed by measuring serum total T4 (TT4; normal range 55-160nmol/L, RAI method, Inep, Zemun) and TSH (normal range 0.15-5mU/L, RAI method, Inep, Zemun). Serum TSH receptor antibodies were measured by the ELISA method (EUROIMMUN Lübeck, Germany; normal range <1U/l).
Statistical analysis
The normality assumption for continuous variables was evaluated by the Kolmogorov-Smirnov test. Continuous variables are presented as means and standard deviations for normally distributed variables or as median and interquartile range (25th to 75th percentiles) for non-normally distributed ones. They were compared using the one-way Student’s t test for paired samples, or its non-parametric equivalent, the Wilcoxon matched-pair signed rank test. Categorical variables are presented as counts and percentages and were compared with the chi-square test or McNemar’s test for repeated measurements. To test parameters independently associated with treatment response, variables (age, gender, smoking, duration of GO and TSHRAb) were entered into the multivariable regression model using the forward stepwise (likelihood ratio) method of entry. For all analyses, a two-sided p<0.05 was considered statistically significant.
RESULTS
The demographic, clinical and biochemical data of the study patients are presented in Table 1. Most of the patients were females (74%), and Graves’ disease was present in 90% of the patients.
Ophthalmologic results
Ophthalmologic measurements before and 6 months after start oftreatment are presented in Figures 1 and 2, and Table 2.
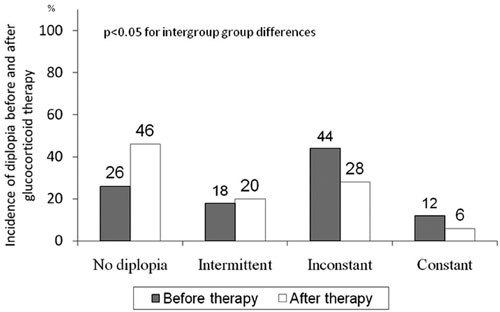
Figure 1. Presence of no diplopia, and intermittent, inconstant and constant diplopia before and after therapy
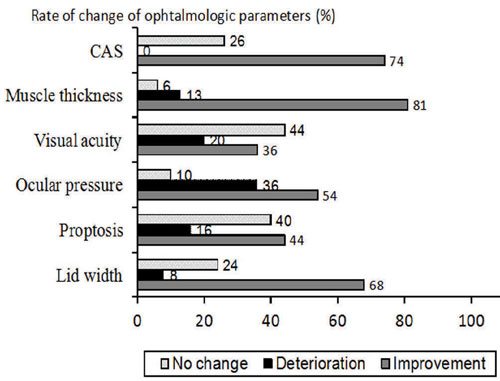
Figure 2. Change in lid width, proptosis, ocular pressure, visual acuity, muscle thickness and CAS before and after therapy.
Diplopia
Before treatment, intermittent, inconstant and constant diplopia was present in 9 (18%), 22 (44%) and 6 (12%) patients, respectively, whereas 13 patients (26%) did not have diplopia. Following treatment, no diplopia was present in 23 patients (46%) while 10 (20%), 14 (28%) and 3 (6%) of the patients had intermittent, inconstant and constant diplopia, respectively (Figure 1) (p<0.001 vs. before treatment). In general, diplopia improved in 21 patients (42%), was unchanged in 28 patients (56%) and deteriorated in 1 patient (2%). Diplopia completely disappeared in 10 (20%) of the patients.
Lid width
Eyelid aperture improved significantly during the course of therapy (Table 2). A decrease in lid width of at least 2mm was observed in 68% of patients, deterioration in 8% of patients and no change in 24% of patients (Figure 2).
Proptosis
There was no significant change in the mean values of proptosis (p=NS) at the end of treatment. Generally, there was improvement in 44% of patients, deterioration in 16% and no change in 40% of patients (Figure 2). However, abnormal exophthalmometer readings (values of ≥19) persisted at the end of follow-up in most of the patients (96%).
Ocular pressure
There was no significant difference in the mean values of intraocular pressure before and after treatment (Table 2). Ocular pressure improved in 54% of patients and deteriorated in 36% of patients, while there was no change in 10% of patients (Figure 2).
Visual acuity
Visual acuity improved in 36% of patients and deteriorated in 20% of patients, while there was no change in 44% of patients (Figure 2).
Muscle thickness
Muscle thickness improved significantly during therapy (Table 2). Improvement was observed in 81% of patients and deteriorated in 13% of patients, while there was no change in 6% of patients (Figure 2).
CAS
Median CAS values decreased from 4.5 to 2.0 (p<0.001). Thirty-seven patients (74%) demonstrated improvement, there was no change in 13 (26%) patients and none of the patients presented with deterioration of inflammatory status (Figure 2).
Overall clinical response
Treatment with glucocorticoids resulted in rapid and clinically significant improvement. According to the defined criteria, 33 out of 50 patients (66%) demonstrated treatment response. During the 6-months follow-up period 2/33 patients (6%) had relapse.
Fifteen of 18 non-smokers (83%) and 18/32 (56%, p=0.067) smokers responded to treatment. Patients with CAS ≥4 points had better treatment response (80%) compared with patients with CAS <4 who had treatment response of 23% (p<0.05). Favorable response was observed in the group with greater TSHRAb(16±16 vs. 6±2, p<0.001). The responders were not different from non-responders with regard to age, gender and duration of hyperthyroidism. According to multivariable regression analysis, the only independent parameter associated with positive treatment response was CAS ≥4 (p<0.001).
The whole group had statistically significant improvement of the QoL score related to visual (physical) function after the treatment, but there was no significant difference between scores related to psychosocial function (Figure 3).
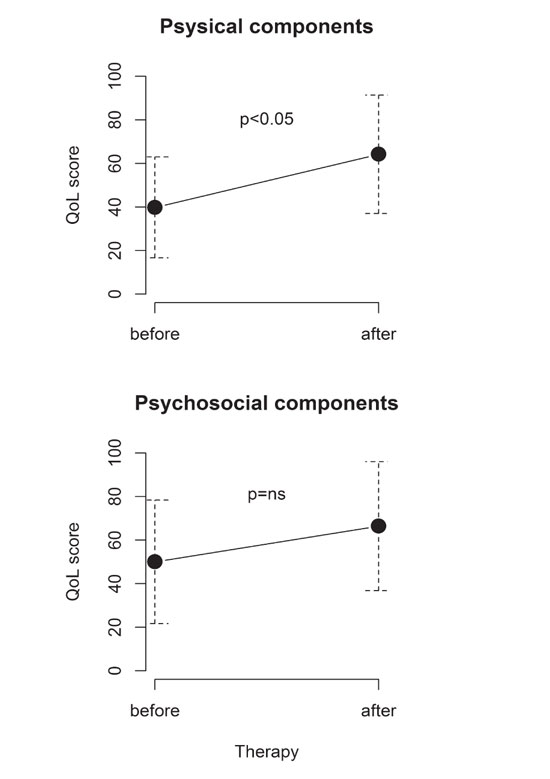
Figure 3. Physical (visual) and psychosocial Quality of life (QoL) assessment before and after treatment.
Adverse events
Mean body weight in the study population increased from 67±12kg to 71±10kg (p<0.001), as did body mass index (24.4±3.8 kg/m2 vs. 26.0±4.0kg/m2, p<0.001). There were no significant changes in blood glucose over 6 months of GC therapy, whereas a significant increase of total cholesterol, LDL-, HDL-cholesterol and tryglicerides was observed (cholesterol: 5.3±1.2 vs. 6.3±1.4mmol/L, p<0.001; HDL-cholesterol: 1.4±0.5 vs. 1.6±0.6mmol/L, p=0.002; LDL-cholesterol: 3.3±1.2 vs. 4.1±1.4mmol/L, p=0.003; tryglicerides: 1.4±0.7 vs. 1.7±0.6mmol/L, p<0.001). In addition, mild, but clinically non-significant, increase in blood pressure was observed (systolic: 122±12 vs. 128±11mmHg, p<0.001; diastolic: 75±6 vs. 80±6mmHg, p<0.001).
The overall rate of steroid-related side effects (Table 3) was 35/50 (70%), but the vast majority of them were mild to moderate and only a few patients developed serious major side effects. In fact, one patient had a marked asymptomatic transitory increase in serum aminotransferases levels without relevant hepatotoxicity, defined as a 4-fold or greater increase in serum liver enzymes and without serologic evidence of viral hepatitis. One patient developed herpes zoster infection and one patient had pulmonary tuberculosis.
DISCUSSION
This was a retrospective analysis of efficacy and tolerability of a combined parenteral and oral glucocorticoid therapy that was applied in 50 euthyroid patients with untreated and active Graves’ orbitopathy. The present study also evaluated the effects of different clinical and laboratory parameters such as smoking, CAS, TSHRAb levels, duration of GO, duration of hyperthyroidism, gender and age impact on the response to glucocorticoids. We have demonstrated that combined parenteral and oral GC therapy is effective and safe in the treatment of Graves’ orbitopathy.
Effectiveness and side effects of GC therapy: Relation to previous studies
The response rate in our study group was 66%. As expected, GC showed the greatest effectiveness on soft tissue changes (incorporated in the CAS), whereas proptosis was less responsive.
Average decrease in CAS of 2.5 and 3.5 points and inactivation of GO according to CAS of 59% and 89% was reported in randomized and nonrandomized trials, respectively.5 Our results demonstrating a significant decrease of 2.5 points are in concordance with previous reports.
Proptosis and lid width did not change significantly in our study and improvement of at least 2mm occurred in 68% of patients, which is in agreement with several previous studies,13,14 while some other studies showed a better response.15,16
In our group of patients the response to GC therapy was influenced by CAS, TSHRAb and smoking behavior. Glucocorticoids have anti-inflammatory and immunosuppressive actions and have favorable effects during the active phase of GO. High CAS, high TSHRAb levels and shorter duration of GO are well known parameters of GO activity and in our study they were predictors of therapeutic efficacy. Smoking affects the immunological reactions in the pathogenesis of the disease. It is liable to alter the structure of thyrotropin receptor making it more immunogenic in a way that leads to the production of thyrotropin receptor stimulating antibodies that react strongly with retro-orbital tissue or, on the other hand, it might impair restoration of tolerance to thyroid auto-antigens.17 In addition, smoking decreases the effectiveness of glucocorticoids in a dose-dependent manner during treatment.18 Our results are in line with other studies and suggest that doctors should urge patients to give up smoking. We did not find any impact of gender and age on the response to therapy, unlike a previous study where patients younger than 50 years of age and female subjects responded better to steroid therapy.16
Adverse events are a major concern of GC therapy with morbidity and mortality rates of 6.5 and 0.6%, respectively.5 There are numerous case series and reports on the potential serious cardiovascular, cerebrovascular and hepatic risks, some even with fatal outcome.5 Severe liver damage was reported after intravenous pulses, probably by a direct toxic effect on hepatocytes, and the extent and outcome seems to be dose-dependent.18 Pretreatment liver steatosis and/or diabetes were not related to liver damage, whereas preexistent viral hepatitis B has been shown to be a risk factor. By contrast, cases of hepatitis and liver failure have not been reported in patients treated with high doses of oral GC, which may be related to the gradual withdrawal consistent with oral therapy. In our study, liver enzymes, evaluated every month, were in the normal range in all patients except one, who had asymptomatic and transitory increase in serum aminotransferases levels with no serologic evidence of viral hepatitis. Severe infections as a consequence of GC immunosuppressive effects have also been documented as serious side effects.19 Likewise, two patients in our group developed serious infections: herpes zoster and pulmonary tuberculosis. In addition, psychosis and major depression have additionally been documented as serious side effects,6 which was not observed in our study group.
All other side effects were non-severe. Weight gain and hirsutismus were the most common, and they occurred more frequently than in other studies, including both IVGC and ORGC therapy, while other mild side effects are in line with other studies. They appeared to be related to cumulative dose (10.2g) and treatment schedule (ORGC+IVGC), whereas serious side effects depend on preexisting disease as well.
In regard to treatment protocol, it is documented that the intravenous route is more effective and better tolerated than the oral route.15,16 However, after discontinuation of pulse therapy, deterioration of eyesight is a fairly common problem. A recent multicenter study6 described relapse in 33%, 21% and 40% corresponding to different cumulative doses of 2.25, 4.98 or 7.47g, respectively, in 12 weekly infusions. Relapse rate in our study was only 6%. It seems that interpulse oral prednisone reduces recurrence rate and maintains improvement stability.
A common protocol uses a 4.5g cumulative dose of methylprednisolone subdivided into 12 weekly infusions in the context of out-of-hospital care16 But in practical terms, it is unfeasible to manage in the same day parenteral therapy (a few hours) and examination, as EUGOGO recommends that all patients with GO, except milder cases, should be referred to a combined thyroid eye clinic in the tertiary center for further assessment and management. Some patients, especially those who live far from a tertiary center and need several hours of travel, preferred our protocol to a weekly protocol and find it more convenient. Thus, the protocol including hospitalization of the patients for 3 days every month, including in-hospital parenteral therapy and oral therapy at home in the meantime, seems more clinically appropriate and feasible. Moreover, considering the possibility of development of side effects, it seems to be safer for the patients to have monthly biochemical and clinical evaluation.
Study Limitations
In comparison to other studies evaluating the effects and safety of different steroids protocols in GO, the present was a single protocol study including a large number of patients. About 35 reports, case series and trials of parenteral GC treatment including a wide range of patients (5-70 patients) was published between 1987 and 2009.5 Thus, our treatment group was one of the largest reported single-center studies, which makes this study particularly relevant in the light of rare diseases such as GO, further confirmed by the recent study from Denmark reporting an incidence of moderate-to-severe and active GO of 15.5/million inhabitants per year.20
CONCLUSION
In conclusion, combined oral and intravenous glucocorticoid therapy is effective, safe and well tolerated, but appropriate selection of patients based on the severity of the disease and the potential for the development of side effects from the high doses of GC therapy is warranted.
CONFLICT OF INTEREST
Νone.
FUNDING
This study was supported partially by grants No. 175036 and No. 175067 of the Ministry of Science of the Republic of Serbia.
REFERENCES
1. Bahn RS, 2010 Graves ophthalmopathy. N Engl J Med 362: 726-738.
2. Burch HB, Wartofsky L, 1993 Graves ophthalmopathy: current concepts regarding pathogenesis and treatment. Endocr Rev 14: 747-793.
3. Bartalena L, Pinchera A, Marcocci C, 2000 Management of Graves ophthalmopathy: reality and perspectives Endocr Rev 21: 168-199.
4. Bartalena L, Baldeschi L, Dickinson AJ, et al, 2008 Consensus statement of the European group on Graves orbitopathy (EUGOGO) on management of Graves orbitopathy. Thyroid 18: 336-346.
5. Zang S, Ponto KA, Kahaly GJ, 2011 Intravenous glucocorticoids for Graves orbitopathy: Efficacy and morbidity. J Clin Endocrinol Metab 96: 320-332.
6. Bartalena L, Krassas GE, Wiersinga W, 2012 Efficiacy of three different cumulative doses of intravenous methylprednisolone for moderate to severe and active Graves’ orbitopathy. J Clin Endocrinol Metab 97: 4454-4463.
7. Kendall-Taylor P, Crombie AL, Stephenson AM, et al, 1988 Intravenous methylprednisolone in the treatment of Graves ophthalmopathy. Br Med J 297: 1574-1578.
8. Dickinson AJ, Perros P, 2001 Controversies in the clinical evaluation of active thyroid associated orbitopathy: use of a detailed protocol with compartice photographs for objective assessment. Clin Endocrinol (Oxf) 55: 283-303.
9. Bahn RS, Gorman C, 1987 Choice of therapy and criteria for assessing treatment outcome in thyroid-associated ophthalmopathy. Endocrinol Metab Clin N Am 16: 391-407.
10. Mourits MP, Prummel MF, Wiersinga WM, Koornneef L, 1997 Clinical activity score as a guide in the management of patients with Graves ophthalmopathy. Clin Endocrinol (Oxf) 47: 9-14.
11. Bartalena L, Marcocci C, Bogazzi F, et al, 1998 Relation between therapy for hyperhyroidism and the course of Graves ophthalmopathy. N Engl J Med 338: 73-78.
12. Terwee CB, Dekker FW, Mourits MP, et al, 2001 Interpretation and validity of changes in scores on the Graves’ ophalmopathy quality of life questionnaire (GO-QoL) after different treatment. Clin Endocrinol (Oxf) 54: 391-398.
13. Kendall-Taylor P, Crombie AL, Stephenson AM, et al, 1998 Intravenous methylprednisolone in the tretment of Graves’ ophthalmopathy. Br Med J 297: 1574-1578.
14. Kaupinen-Makelin R, Karma A, Leinomen E, et al, 2002 High dose intravenous methylprednisolone puls therapy versus oral prednisone for thyroid associated ophthalmopathy. Ophthalmol Scand 80: 316-321.
15. Marcocci C, Bartalena L, Tanda LM, et al, 2001 Comparison of the effectiveness and tolerability of intravenous or oral glucocorticoids associated with orbital radiotherapy in the management of severe Graves’ ophthalmopathy: results of a prospective, single-blind, randomized study. J Clin Endocrinol Metab 86: 3562-3567.
16. Kahaly GJ, Pitz S, Hommel G, Dittmar M, 2005 Randomized, single blind trial of intravenous versus oral steroid monotherapy in Graves’ orbitopathy. J Clin Endocrinol Metab 90: 5234-5240.
17. Utiger RD, 1998 Effects of smoking on thyroid function. Eur J Enocrinol 138: 368-369.
18. Hegedius L, Brix TH, Vestergaard P, 2004 Relationship between cigaretes smoking and Graves ophthalmopathy. J Endocrinol Invest 27: 265-271.
19. Le Moli R, Baldeschi I, Saeed P, Regensburg N, Mourits MP, Wiersinga WM, 2007 Determinants of liver damage associated with intravenous methylprednisolone pulse therapy in Graves ophthalmopathy. Thyroid 17: 357-362.
20. Laurberg P, Berman DC, Bülow Pedersen I, Andersen S, Carlé A, 2012 Incidence and clinical presentation of moderete to severe Graves’ Orbitopathy in a Danish population before and after iodine fortification of salt. J Clin Endocrinol Metab 97: 2325-2332.
Address for correspondence:
Assist. Prof. Biljana Nedeljkovic Beleslin, MD, PhD, Clinic for Endocrinology, Diabetes and Metabolic Disorders, Clinical Center of Serbia, Dr Subotica 13, Belgrade, Tel./Fax: +381113639723, E-mail: biljana_beleslin@yahoo.com
Received 07-04-2013, Accepted 05-08-2013