Abstract
OBJECTIVE: the aim of this study was to search for a possible association of low bone mineral density (bMD) with the PvuII and XbaI polymorphisms of the estrogen receptor (Er) gene in bulgarian women. DESIGN: 400 bulgarian women participated in this study. bMD was mea-sured at the lumbar spine, femoral neck and at the distal forearm. two groups were identified: women with normal bMD at both central sites (n=180) and women with low bMD at either site (n=220), designated as normal (NbMD) and low bMD (LbMD) groups, respectively. the genotype frequencies of PP, Pp, pp and XX, Xx, xx were investigated by Pcr and enzymatic digestion of the products by PvuII and XbaI. RESULTS: the genotype frequencies were 12% for the PP, 59% for the Pp and 29% for the pp genotypes in the NbMD, and 26%, 50% and 24% in the LbMD groups, respectively. the XX, Xx, xx genotype frequencies were 14%, 63% and 23% in the NbMD, and 33%, 50% and 17% in the LbMD groups, respectively. the various genotypes were significantly associated with bMD. the relative risk for low bMD was higher for the XbaI marker (rr=1.51) than for the PvuII marker (rr=1.35). the association between low bMD and the polymorphisms under study was described by an etiological factor of 0.28 for the XbaI marker and 0.20 for the PvuII marker. CONCLUSIONS: the specific XbaI and PvuII polymorphisms of the Er gene are associated with low bMD at all bMD measurement sites in the bulgarian female population. they might therefore become useful genetic markers in osteoporosis risk assessment in this specific population.
Osteoporosis is a systemic skeletal disease characterized by low bone mass and micro-architectural deterioration of bones, leading to increased bone fragility and a consequent increase in fracture risk. Estrogens are an important factor maintaining bone mass in women.1 Polymorphisms in the estrogen receptor (ER) gene have been extensively studied for several years and various polymorphic regions with a significant impact on bone mass and bone metabolism have been identified. A study by Kobayashi et al showed that the PvuII and XbaI polymorphisms of the ER gene were associated with different levels of bone mineral density (BMD) in the lumbar spine in post menopausalJ apanese women.2 A similar study failed to detect significant differences in BMD among ER genotypes in postmenopausal women on hormone replacement therapy (HRT).3 The former study suggested that ER polymorphisms could be related to the acquisition of peak bone mass,whereas the latter hypothesized an influence on the rate of bone loss. Another study on premenopausal and late postmenopausal Japanese women indicated a clear association between the ER genotypes and peak bone mass in adulthood, an association which weakened with advancing age.4 Willing et al found a significant association between the PvuII or XbaI genotypes and the lumbar spine BMD in 253 pre- and perimenopausal white women5. Carriers of the pp genotype had 6.4% lower BMD than women with the PP genotype.5 Over a 3-year period there were no differences in BMD losses or in biochemical markers of bone turnover between the two groups. A study investigating the 5-year bone loss in early postmenopausal women on HRT or placebo could not find significant differences in BMD attributable to the ER polymorphisms at baseline. Nevertheless, in the absence of HRT, significant differences in lumbar spine BMD losses were registered in carriers of the ER genotypes PP (-6.4%) and pp (-2.9%),6 a finding not detected in women on HRT.
Recent studies have shown that estrogens may influence bone formation in men. Riggs et al suggested that estrogen deficiency might also contribute to longitudinal bone loss in men.7 An association between ER gene polymorphisms and lumbar spine BMD was demonstrated in a group of men aged 20 to 79 years.8 Smith et al described osteoporosis in a man with estrogen resistance due to mutation of the ER gene.9
The ER genotype predictable of low or high BMD may be different in the various populations. Thus, the ER PvuII (-/-) genotype was associated with low BMD in Japanese and Italian women,10,11 while the PvuII (+/+) genotype was associated with low BMD in American and Finnish women.12,13 ER polymorphisms have also been associated with an increased risk for osteoarthritis.14
We conducted a study in a population sample of 400 Bulgarian women looking for a possible associa-tion between BMD and PvuII and XbaI polymor-phisms of the ER gene.
SUBJECTS AND METHOD
Subjects
Four hundred unrelated women of Bulgarian origin were recruited for this study. They had been referred for bone densitometry by General Practitioners or other medical specialists or were self-referrals because of their concern about possible osteoporosis. Diseases and medications known to affect bone metabolism were used as exclusion criteria. All participants gave their informed consent before enrollment. The age of the participants ranged from 46 to 65yrs and they were classified according to BMD values into two groups: Normal BMD group (NBMD, n=180) and low BMD group (LBMD, n=220). All studied women were in menopause.
This work was approved by the Ethics Committee at the Alexandrovska Hospital.
Bone densitometry
BMD was measured at the distal forearm by single-energy X-ray absorptiometry (SXA) on a DTX-100 Unit (Osteometer Meditech, USA) and at the lumbar spine (L1-L4) and femoral neck by dual-energy X-ray absorptiometry (DXA) on a Hologic QDR 4500 A device with software version 8.26:3 (Hologic Inc., Waltham, MA, USA).
On the DTX-100 the distal region of interest begins at the 8mm separation point between radius and ulna and then continues proximally for a distance of 24mm. The ultra-distal site extends from the radial endplate proximally to the 8mm point. BMD was measured according to the manufacturer’s instructions in g/cm2 separately for the distal (including radius and ulna) and the ultra-distal site (including only the radius). T-and Z-scores were calculated automatically using the reference values based on the manufacturer’s Danish database (issued 1994).
Lumbar spine and femoral neck BMD were ex-pressed in g/cm2 and additionally in terms of age-matched Z-scores. The manufacturer’s American reference database including the NHANES III da-taset (for the femoral neck) was used.
All subjects were grouped according to their BMD. Two hundred and twenty participants had low BMD (osteopenia or osteoporosis) at either the lumbar spine or femoral neck (low density group –LBMD-) and 180 had normal BMD at both central sites (normal density group -NBMD-).
Genotyping
The DNA was isolated from whole blood. The primers and PCR conditions for amplifying intron 1 of the ER gene were designed according to the procedures described by Lorentzon et al.15 dATP, dCTP, dTTP, dGTP – 1.25mM each were used to amplify intron 1 with Taq DNA Polymerase. 100ng of the DNA were used as template in the PCR reactions.
The first polymorphic region under study is located in the long arm of chromosome 6 (6q25,1). Forward and reverse primers were used to amplify the PCR product which was digested with PvuII. The digestion of the PCR product with PvuII generated two fragments. Individuals homozygous for the PP genotype had a single uncut fragment, while those homozygous for the pp genotype had two fragments. The heterozygotes Pp had all three bands.
The second polymorphic region under study is located in 6q25,1. Forward and reverse primers were used to amplify the PCR product which was then digested with XbaI. XbaI cuts the x allele of the ER gene but not the X allele. Digestion of the PCR product with XbaI generated two fragments. Individuals homozygous for the XX genotype had a single uncut fragment, while those homozygous for the xx genotype had two fragments. The heterozygotes Xx had all three bands.
The PCR products were digested with PvuII and XbaI for 4h and electrophoresed through a 2% agarose gel. Individuals were scored as PP, Pp, pp and XX, Xx, xx according to their digestion pattern. Uppercase letters mark the absence and lowercase letters mark the presence of restriction sites.
The PCR quality control was provided by blank probe (negative control) containing all reaction components except DNA. For the accuracy of the restriction analysis a positive control of the molecular marker 100 bp was used.
Statistical analysis
Data were evaluated by the Χ2-test and presented as mean ± SD.
The relative risk (RR) for low BMD in the presence of a specific allele was calculated as RR = (a x d)/(b x c), where a, b, c and d are the numbers of car-riers/non-carriers in the low density group (LBMD)/ normal density group (NBMD) as shown below:
| Carriers | Non-carriers | |
| LBMD | a | B |
| NBMD | c | D |
The etiological factor (EF) showing what part of the condition (low BMD) might be attributable to the associated factor (ER gene polymorphisms) on a population level was calculated as EF = (RR-1)/RR = a/(a+b), where a is the number of carriers among the LBMD and b is the number of non-carriers among the LBMD.
RESULTS
The PvuII marker
The observed distribution of the different geno-types and alleles according to the PvuII in the two groups is shown in Table 1 .
The allelic and genotype frequencies were calculated in LBMD (low BMD and/or osteoporosis) and in NBMD (normal BMD), and in the homo-and heterozygotes (Table 1). The results were statisti-cally significant (Χ2-test, p<0.05). When comparing the genotype frequencies in LBMD and in NBMD, the PP was more common in LBMD (26%) than in NBMD (12%). In contrast, the pp was less common in LBMD (24%) than in NBMD (29%). The frequency of heterozygotes was higher in the NBMD (59%) than in the LBMD (50%). The differences reached statistical significance (Χ2-test, p<0,05). The marker is characterized by high polymorphism information content (PIC) of 0.37 and a high part of heterozygotes (H0) (0.50) (Tables 1, 2).
The three studied genotype groups (PP, Pp, pp) in NBMD and LBMD do not differ in age, years since menopause, number of smokers and alcohol consumers (Table 2 ).
The distribution of BMD at various sites among the Pvull genotypes is shown in Table 2 and Figure 1. Higher BMD values were found in pp individuals and lower ones in PP individuals. The correlations remained similar for calculated T-and Z-scores also (Figure 1).
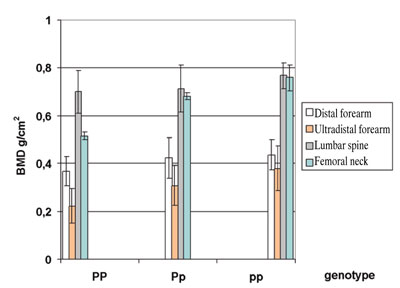
Figure 1.BMD (g/cm2) of the distal and ultradistal forearm, lumbar spine (L1-L4) and femoral neck for the different PvuII genotypes. Data are shown as mean ±SD.
Student’s t-test:Distal forearm site: p=n.s. for pp vs. Pp, p=0.05 for pp vs. PP; Ultradistal forearm site: p=n.s. for pp vs. Pp, p<0.01 for pp vs. PP;
Lumbar spine p=n.s. for pp vs. Pp, p=0.04 for pp vs. PP;
Femoral neck: p=0.03 for pp vs. Pp, p<0.001 for pp vs. PP.
The associations of the different PvuII genotypes with the BMD in the lumbar spine and femoral neck are shown in Table 2 . The associations of the lumbar spine and femoral neck T-and Z-scores with the PvuII genotype resembled those with the forearm T- and Z-scores (Figure 1).
The relative risk (RR) for low BMD in the pres-ence of the PvuII marker is 1.35. The etiological factor (EF), which shows what part of the illness at populational level is due to the marker PvuII, is 0.20.
The XbaI marker
The observed distribution of the different genotypes and alleles in the LBMDand NBMDaccording to the XbaI marker is shown in Table 3.
The allelic and genotype frequencies by XbaI in the LBMD (with low BMD and/or osteoporosis) and in the NBMD, and in the homo- and heterozygotes, are presented in Table 3. The results were statistically significant by applying the X2-test (p<0.05).The XX genotype was more frequent in LBMD with low BMDand/orosteoporosis (33%) than in NBMD with normal BMD (14%). By contrast, the xx genotype was less frequent in LBMD (17%) than in NBMD (23%) (Table 3). The heterozygotes Xx were more often seen in NBMD (63%) than in LBMD (50%). The differences reached statistical significance by applying the X2-test (p<0.05). The marker is characterized with high polymorphism information content (PIC) and a high part of heterozygotes (H0) (Tables 3, 4).
The three studied genotype groups (XX, Xx, xx) in NBMD and LBMD do not differ in age, years since menopause, number of smokers and alcohol consumers (Table 4).
The distribution of BMD at various sites and otherpertinentparametersamongthedifferentXbal genotypes are shown in Table 4. The introduction of T-and Z-scores did not change the tendency (Figure 2). The distribution of BMD with the genotype was similar at all three measured sites. Lumbar spine and femoral neck T-scores were higher in xx and lower in XX individuals (Figure 2). The RR for low BMD in the presence of the XbaI marker is 1.51. The EF of the marker XbaI is 0.28.
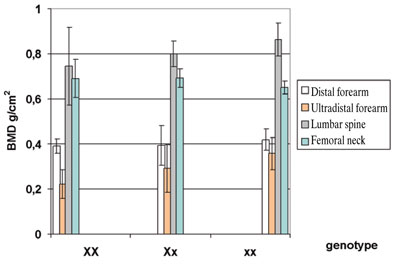
Figure 2.BMD (g/cm2) of the distal and ultradistal forearm, lumbar spine (L1-L4) and femoral neck for the different XbaI genotypes. Data are shown as mean ±SD.
Student’s t-test:Distal forearm site: p=n.s. for xx vs. Xx, p=0.06 for xx vs. XX; Ultradistal forearm site: p=n.s. for xx vs. Xx, p=0.02 for xx vs. XX; Lumbar spine: p=n.s. for xx vs. Xx, p=0.04 for xx vs. XX; Femoral neck: p=n.s. for xx vs. Xx, p=n.s. for xx vs. XX.
DISCUSSION
The molecular mechanisms underlying the in-fluence of PvuII and XbaI polymorphisms of the estrogen receptor gene on bone mass are poorly understood. The PvuII and XbaI polymorphisms are located in a non-functional area of the ER gene but in linkage disequilibrium with the TA polymorphism in the ER promoter. It is thus likely that the TA repeated sequence could alter gene transcription, though this has not yet been elucidated.10,11
The genetic variance of the ER locus contributes to the maintenance of peak bone mass and predisposes some individuals to rapid bone loss and osteoporosis. The precise mechanisms by which variance of the ER locus reduces bone quantity and quality leading to osteoporotic fractures are currently under investigation. The observed associations of the P allele of the PvuII site and the X allele of the XbaI with low BMD indicate that the ER gene is involved in the pathogenesis of osteoporosis. The higher proportion of Pp and Xx in our subjects with normal BMD suggests a lack of effect of the heterozygous state on the predisposition to low BMD. Contrary to our data, a study conducted in an Italian cohort indicated that the PP and XX genotypes were associ-ated with higher bone density, as was also reported forThailandandChinesepopulations.10,11 Ourresults are in line with those from the Finnish population, in which the pp and xx genotypes were associated with slower rates of bone loss and consequently higher bone density.13 The differences observed among the various population groups cannot be interpreted at present.
With regard to the Pvull polymorphisms, comparing our Bulgarian population under study with pub-lished data of women of European and Asian origin, we observed a clear difference in the distribution of the alleles frequencies. For the purpose of this comparison we used the published genotype frequencies for menopausal European (18% for the PP, 53% for the Pp and 29% for the pp genotype) and Asian women (18% for the PP, 52% for the Pp and 30% for the pp genotype).16 Regarding the association of the genotype with the presence of a low BMD (LDG), our data are in line with already published data but a major difference is seen in the PP genotype.
With regard to the XbaI polymorphisms, the observed alleles frequencies in our study population (LBMD, n=220) differed significantly from those detected in other European (10% for the XX, 48% for the Xx and 42% for the xx genotype) and Asian menopausal populations (5% for the XX, 31% for the Xx and 64% for the xx genotype).16 The major differences were seen in the XX and xx sub-groups. The reasons for this variability are not apparent.
In our study, individuals with the PP genotype (12% of the NBMD and 26% of the LBMD) had 11% lower forearm BMD than the subjects with the pp genotype (29% of the NBMD and 24% of the LBMD). The Pp heterozygotes (59% of the NDG and 50% of the LBMD) had intermediate BMD values. The association of BMD with the genotype was also confirmed in the lumbar spine and femoral neck. In the pp genotype the reduction in lumbar spine BMD exceeded that in forearm BMD (-27% vs. -5%). In the PP genotype the reduction in fore-arm BMD exceeded that seen in lumbar spine BMD (-33% vs. -36%). At the femoral neck, the highest BMD reduction was seen in the PP genotype (-45%). The difference between the two genotypes pp and PP is statistically significant for BMD measured at distal forearm (consisting of cortical bone) and ultradistal forearm (consisting of trabecular bone). The difference reaches statistical significance for pp and PP BMD of lumbar spine (trabecular bone) and for pp, Pp, PP BMD of femoral neck (cortical bone). These data show the probable role of the PvuII polymor-phism for trabecular and for cortical bone.
Women with the XX genotype (14% of the NBMD and 33% of the LBMD) had 18% lower forearm BMD than subjects with the xx genotype (23% of the NBMD and 17% of the LBMD). The Xx heterozygotes (63% of the NBMD and 50% of the LBMD) had intermediate BMD values. The same association of BMD with the genotype was seen in the lumbar spine and femoral neck. The BMD reduction was lower than that at the forearm (-29% and -27% vs. -35%) in XX subjects. The BMD reduction was lower than that at the femoral neck (-17% vs. -25%) in xx individuals. The difference between the two genotypes xx and XX is statistically significant for BMD measured at ultradistal forearm (trabecular bone) but not at distal forearm. The difference reaches statistical significance for xx and XX BMD of the lumbar spine (trabecular bone). There is no statistical significance for BMD of the femoral neck. These data show the probable role of the XbaI polymorphism for trabecular bone but not for cortical bone.
The RR for low BMD (osteopenia/osteoporosis) in the presence of the PvuII marker was 1.35 and 1.51 in the case of the XbaI marker. The RR for low BMD was higher for the XbaI than for the PvuII marker. The risk of low BMD was higher in X car-riers than in P carriers. The EF, which shows what part of the condition is attributable to the associated factor on a population level, was found to be 0.20 for the PvuII marker and 0.28 for the XbaI marker. Our data showed that a higher proportion of low BMD was attributable to the XbaI marker (28%) than to the PvuII marker (20%). This fact suggested that the XbaI marker was more informative from a clinical point of view.
In conclusion, this is the first study examining the ER gene polymorphisms in Bulgarian women. We found that the prevalence of the PvuII polymor-phisms was similar to that published for other European and Asian populations, whereas the prevalence of the XbaI polymorphism was different than that in the same reference populations. The impact of the different genotypes according to the PvuII marker was confirmed in the forearm site, the lumbar spine and the femoral neck and in the forearm and the lumbar spine according to the XbaI marker. Our data underline the potential benefit of screening subjects at risk for genetic predisposition for osteoporosis.
REFERENCES
1. Lufkin EG, Wahner HW, O’Fallon WM, et al, 1992 Treatment of postmenopausal osteoporosis with transdermal estrogen. Ann Intern Med 117: 1-9.
2. Kobayashi S, Inoue S, Hosoi T, Ouchi Y, Shiraki M, Orimo H, 1996 Association of bone mineral density with polymorphism of the estrogen receptor gene. J Bone Miner Res 11: 306-311.
3. Han KO, Moon G, Kang YS, Chung HY, Min HK, Han IK, 1997 Non-association of estrogen receptor genotypes with bone mineral density and estrogen responsiveness to hormonereplacementtherapyinKoreanpostmenopausal women. J Clin Endocrinol Metab 82: 991-995.
4. Mizunuma H, Hosoi T, Okano H, et al, 1997 Estrogen receptor gene polymorphism and bone mineral density at the lumbar spine of pre-and postmenopausal women. Bone 21: 379-383.
5. Willing M, Sowers M, Aron D, et al, 1998 Bone mineral density and its change in white women: estrogen and vitamin D receptor genotypes and their interaction. J Bone Miner Res 13: 695-705.
6. Salmen T, Heikkinen AM, Mahonen A, et al, 2000 Early postmenopausal bone loss is associated with PvuII estrogen receptor gene polymorphism in Finnish women: effect of hormone replacement therapy. J Bone Miner Res 15: 315-321.
7. Riggs L, Khosla S, Melton J, 1998 A unitary model for involutional osteoporosis: estrogen deficiency causes both type I and type II osteoporosis in postmenopausal women and contributes to bone loss in aging men. J Bone Miner Res 13: 763-773.
8. Ongphiphadhanakul O, Rajatanavin R, Chanprasertyothin S, Piaseu N, Chailurkit L, 1998 Serum oestradiol and oestrogen – receptor gene polymorphism are associ-ated with bone mineral density independently of serum testosterone in normal males. Clin Endocrinol (Oxf) 49: 803-809.
9. Smith EP, Boyd J, Frank GR, et al, 1994 Estrogen re-sistance caused by a mutation in the estrogen receptor gene in a man. N Engl J Med 331: 1056-1061.
10. Gennari L, Becherini L, Masi L, et al, 1998 Vitamin D and estrogen receptor allelic variants in Italian postmenopausal women: Evidence of multiple gene contribution to bone mineral density. J Clin Endocrinol Metab 83: 939-944.
11. Kobayashi S, Inoue S, Hosoi T, Ouchi Y, Shiraki M, Orimo H, 1996 Association of bone mineral density with polymorphisms of the estrogen receptor gene. J Bone Miner Res 11: 306-311.
12. Langdahl BL, Lokke E, Carstens M, Eriksen EF, 1997 Polymorphisms in the estrogen receptor gene show different distributions in osteoporotic patients and normal controls. J Bone Miner Res 12: S255.
13. Mahonen A, Turunen AM, Kroger H, Maenpaa PH, 1997 Estrogen receptor gene polymorphism is associated with bone mineral density in perimenopausal Finnish women. J Bone Miner Res 12: T614.
14. Ushiyama T, Ueyama H, Inoue K, Nishioka J, Ohkubo I, Hukuda S, 1998 Estrogen receptor gene polymorphism and generalized osteoarthritis. J Rheumatol 25: 134-137.
15. Lorentzon M, Lorentzon R, Backstrom T, Nordstrom P, 1999 Estrogen receptor gene polymorphism, but not estradiol levels, is related to bone density. J Clin Endocr Metab 84: 4597-4601.
16. Rizzoli R, Bonjour J-P, Ferrari SL, 2001 Osteoporosis, genetics and hormones. J Molec Endocrinol 26: 79-94.
Address for correspondence:
M.A. Boyanov, MD, PhD, Endocrinology Clinic,
Alexandrovska Hospital, 1 G. Sofiiski Str., Sofia 1431,
Bulgaria (Europe); Phone: +3592 9230 784,
Fax: +3592 9230 779,
E-mail: boyanov@alexandrovska-hospital.bg
mihailboyanov@yahoo.com
Received 01-03-06, Revised 26-10-06, Accepted 10-11-06