Abstract
Ectopic production of CRH by a medullary thyroid carcinoma or its metastases is a rare cause of ectopic Cushing’s syndrome (ECS). We report a 45-year old male with medullary thyroid carcinoma (MTC), who, 24 years following the initial diagnosis, presented with clinical and biochemical evidence of an ACTH dependent Cushing’s syndrome. Rapid deterioration of his clinical condition and elevated cortisol levels were observed. Computed tomographic imaging of the abdomen revealed extensive liver metastases. The patient underwent fine needle aspiration biopsy of a liver lesion and immunohistochemistry showed that the cells expressed calcitonin, carcino-embryonic antigen and synaptophysin. Further analysis revealed that the material also expressed CRH. This is an unusual case of a CRH-secreting liver metastasis from a medullary thyroid carcinoma 24 years after the initial diagnosis of MTC.
PATIENT’S DESCRIPTION
A 45-year old man was admitted to the Endocrine Unit in April 2005 complaining of fatigue and leg weakness of two weeks duration. Clinical examination revealed a moon face, proximal myopathy, edema of the lower extremities and mild hypertension.
Laboratory investigations at that time demonstrated fasting blood glucose of 11.1 mmol/L (normal: 4.2-6.4 mmol/L), potassium of 2 mmol/L (normal: 3.5-5.0 mmol/L), total bilirubin of 51 mmol/L (normal: 3-17 mmol/L) and impaired liver function tests.
The patient had a past history of medullary thyroid carcinoma (MTC) diagnosed 24 years previously. He was first operated on in 1981 (total thyroidectomy and removal of an affected lymph node of the right neck). He was re-operated on in 1990 and also in 1993 (for lymph nodes dissection of the right neck). Genetic testing (PCR amplification and direct sequencing) did not disclose RET mutations. The family history was negative for MTC and other NET tumors as well as for other chronic diseases. It was assumed that the patient had a sporadic MTC. Because of residual disease and biochemical deterioration, the patient received chemotherapy with paraplatin and etoposide (8 cycles) in 1996. During close follow-up, abdominal computed tomography (CT) did not show any pathology in the liver or pancreas. Whole body scans with 111In-[D-Phe]-DTPA-octreotide and [131I]-metaiodobenzylguanidine (MIBG) were also negative. 111In-[D-Phe]-DTPA-octreotide scan were positive in 1999 and MIBG scan in 2001, both indicating tracer activity in the hilum of the left lung. Thorax CT in 2000 indicated the presence of small metastatic lung lesions in both lungs. Between 1999-2001, the patient received therapy with long-acting somatostatin analogues for the control of symptoms (mainly flushing). In 2002, when MIBG treatment became available in our hospital, the patient received therapy with two doses of MIBG (total dose of 300 mCi). Post therapeutic scan carried out after the first and second dose showed uptake in the hilum of the left lung. In the 2nd post therapeutic scan the uptake was significantly lower than in the 1st post therapeutic scan or in the diagnostic scan of 2001. Calcitonin levels remained elevated, despite treatment, ranging from 1000 to 1500 ng/L (normal: below 10 ng/L).
Despite lack of biochemical improvement, the patient had been kept in good general condition until April 2005, when he started complaining of persistent muscle weakness. Because of the clinical manifestations of severe proximal myopathy and hypokalemia, which could not be attributed to his cancer (lack of diarrhea or vomiting), the existence of a second pathology, such as Cushing’s syndrome, was suspected. Hormonal analysis revealed elevated serum morning cortisol 1504 nmol/L (normal: 150-650 nmol/L) and plasma ACTH of 81 pmol/L (normal: 2-11.5 pmol/L). Chromoganin A was mildly elevated (5.6 ng/ml, normal: 1.5-5 ng/ml). Overnight administration of 8 mg of dexamethasone did not suppress cortisol levels, which remained elevated at 765 nmol/L on the following morning sample.
A pituitary magnetic resonance imaging (MRI) was normal. An abdominal CT showed normal adrenals, but multiple liver lesions. As the diagnosis of ectopic Cushing’s syndrome (ECS) was raised, the parallel evolution of hypercortisolemia and growth of liver lesions implied that ECS was caused by the hepatic lesions (most likely metastastic). The patient underwent biopsy of the larger liver mass. Immunohistochemistry showed that the cells expressed calcitonin, carcino-embryonic antigen (CEA) and synaptophysin, suggestive of a hepatic metastasis from the known MTC (Figure 1). Material from the biopsy, also examined by immunohistochemistry, showed strong reactivity for CRH (Figure 2), while immunostains were negative for ACTH.
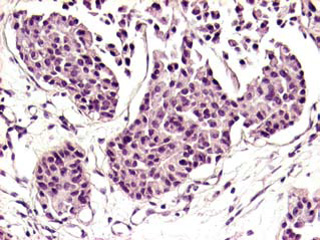
Figure 1. Liver metastasis of MTC composed of solid nests. The medium-sized tumor cells harbor ovoid, moderate hyperchromatic nuclei, focally with conspicuous nucleolus (H&e 20x).
The patient was started on metyrapone (250 mg 3 times daily), potassium supplements, insulin and low-weight heparin. Unfortunately, he could not tolerate a higher metyrapone dose. The patient’s clinical condition deteriorated and he refused any further treatment. He left the hospital and died at home three months later. Autopsy was not performed.
METHODS
ACTH was polyclonal, dilution 1:2000, provided by the NHPP (National Hormone Pituitary Program). ACTH-secreting adenomas were used as positive controls.
CRH was polyclonal, dilution 1:100, provided by Peninsula Lab, Belmont CA. Ectopic CRH-secreting endocrine tumors of the lung were used as positive controls. All tissue control samples were retrieved from the files of the Department of Pathology and Pituitary Tumor Reference Center, G. Gennimatas, Athens General Hospital.
DISCUSSION
MTC may be associated with a variety of humorally mediated syndromes. Clinical manifestation of ECS is a rare event in patients with MTC, seen in 0.6% of patients,1 and has been described in sporadic and familial cases. Among patients with ECS, MTC represents 2.2-7.5% of cases.1-3 The clinical course of the disease is usually short and the prognosis unfavorable, although survival depends on the type of the initial tumor.2 Diagnosis is, in some cases, very difficult and ECS is confirmed by a combination of tests and imaging modalities.2 The diagnosis is based on hypercortisolism with normal or elevated levels of plasma ACTH, usually not suppressible by high doses of dexamethasone, and absence of other known causes of CS (as pituitary adenoma).4 Since there is no single accurate test for the differential diagnosis of ACTH dependent CS, a variety of diagnostic algorithms have been used.3,5 Immunohistochemistry and in situ detection of ACTH and/or CRH mRNA may prove helpful in some cases, although production of ACTH precursors by extrapituitary tumors may cause negative immunostains.6 In our case, the metastatic deposits were conclusively positive for calcitonin, CEA and synaptophysin, results in keeping with the diagnostic criteria of MTC according to the recent WHO classification.7 CRH expression was also demonstrated in the metastatic liver lesions. We did not measure plasma CRH, which is elevated in cases of ectopic CRH secretion,8 as this estimation was not available at our hospital. Imaging did not demonstrate pituitary and adrenal hyperplasia in this patient, as expected, and this probably relates to: a) the fact that imaging took place early in the course of disease and b) the rapid course of ECS in this patient.
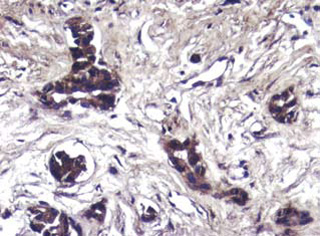
Figure 2. The cells cluster of metastatic MTC show strong immunoreactivity for CRH (Immunostain 20x).
Symptoms in patients with ECS may slightly differ between patients with small-cell carcinomas and those with neuroendocrine tumors.2 In the latter group, weakness, myopathy, hypertension along with hyperglycemia and hypokalemia appear in most patients,9 as was also observed in our patient. Since hypercortisolemia rapidly deteriorates the clinical condition of the patient, it is very important to restore eucortisolemia as soon as possible.
In most cases, MTC diagnosis precedes that of ECS.4 As tumors acquire the ability to produce ACTH and/or CRH at different stages of the disease, variable periods have been reported between the onset of MTC and ECS. The site of ACTH and/or CRH production may be the primary tumor10 or its metastases.11 In our case, the patient developed ECS 24 years after the diagnosis of MTC. To our knowledge, there is only one other report of a patient with a MTC developing ECS 24 years after the diagnosis of cancer,4 which was, however, of uncertain immunohistochemical confirmation. Immunostaining for ACTH/CRH in the initial thyroid tumor of our patient was not possible, since thyroidectomy was performed in 1981 and malignant thyroid tissue was not available.
The optimal therapy for Cushing’s caused by extra-adrenal tumors is the surgical removal of the tumor, although in many cases this is not visible. Adrenolytic medications and bilateral adrenalectomy (nowadays laparoscopic adrenalectomy) have been used to control hypercortisolemia.12 In our case, the patient refused to be treated medically or surgically.
In our patient, the exact cause of death is not known, since he left the hospital against medical advice and died at home. In this group of patients, infections and thrombotic episodes are frequent causes of death.3,13
In conclusion, this case illustrates the rare appearance of ECS in a patient with MTC 24 years after the initial diagnosis, caused by a CRH secreting liver metastasis. To our knowledge, such a long interval between MTC diagnosis and onset of ECS from a metastasis expressing CRH is unique.
REFERENCES
1. Laboureau-Soares Barbosa S, Rodien P, Leboulleux S, et al, 2005 Ectopic Adrenocorticotropic Hormone-Syndrome in Medullary Carcinoma of the Thyroid: A Retrospective Analysis and Review of the Literature. Thyroid 15: 618-623.
2. Isidori AM, Kaltsas GA, Pozza C, et al, 2006 The ectopic Adrenocorticotropin Syndrome: Clinical Features, Diagnosis, Management, and Long-term Follow-up. J Clin Endocrinol Metab 91: 371-377.
3. Ilias I, Torpy DJ, Pacak K, Mullen N, Wesley RA, Nieman LK, 2005 Cushing’s Syndrome due to Ectopic Corticotrophin Secretion: Twenty Years’ Experience at the National Institutes of Health. J Clin Endocrinol Metab 90: 4955-4962.
4. Aniszewski JP, Young WF Jr, Thompson GB, Grant CS, van Heerden JA, 2001 Cushing syndrome due to ectopic adrenocorticotropic hormone secretion. World J Surg 25: 934-940.
5. Al-Saadi N, Diederich S, Oelkers W, 1998 A very high dose dexamethasone suppression test for differential diagnosis of Cushing’s syndrome. Clin Endocrinol 48: 45-51.
6. Smallridge RC, Bourne K, Pearson BW, van Heerden JA, Carpenter PC, Young WF, 2003 Cushing’s syndrome due to medullary thyroid carcinoma:diagnosis by proopiomelanocortin messenger ribonucleic acid in situ hybridization. J Clin Endocrinol Metab 88: 4565-4568.
7. De Lellis LA, Heitz P, Lloyd RV, Eng C (eds) 2004 WHO Classification of Tumours of the Endocrine Organs: Pathology and Genetics of Endocrine Organs, IARC Press, Lyon; p, 40.
8. Wajchenberg BL, Mendonca BB, Liberman B, et al, 1994 Ectopic adrenocorticotropic hormone syndrome. Endocr Rev 15: 752-787.
9. Bayraktar F, Kebapcilar L, Kocdor MA, et al, 2006 Cushing’s syndrome due to ectopic CRH secretion by adrenal pheochromocytoma accompanied by renal infarction. Exp Clin Endocrinol Diabetes 114: 444-447.
10. Jolivet J, Beauregard H, Somma M, Band P, 1980 ACTH-secreting medullary carcinoma of the thyroid: monitoring of clinical course with calcitonin and cortisol assays and immunohistochemical studies. Cancer 46: 2667-2670.
11. Von Mach MA, Kann P, Piepkorn B, Bruder S, Beyer J, 2002 Cushing’s syndrome caused by paraneoplastic ACTH secretion 11 years after occurrence of a medullary thyroid carcinoma. Dtsch Med Wochenschr 127: 850-852.
12. Mure A, Gicquel C, Abdelmoumene N, et al, 1995 Cushing’s syndrome in medullary thyroid carcinoma. J Endocrinol Invest 18: 180-185.
13. Sarlis NJ, Chanock SJ, Nieman LK, 2000 Cortisolemic indices predict severe infections in Cushing syndrome due to ectopic production of adrenocorticotropin. J Clin Endocrinol Metab 85: 42-47.
Address for correspondence:
Dr Alexandra Chrisoulidou, Unit of Endocrinology & Endocrine Oncology, “Theagenio” Hospital,
2 Al. Simeonidi Str., 540 07 Thessaloniki, Greece, Tel: 2310 898 681, Fax: 2310 898 809,
e-mail: alexri@otenet.gr
Received 18-12-07, Revised 15-05-08, Accepted 10-06-08