Abstract
A patient with an intrasellar germinoma leading to pituitary stalk thickening is reported. The patient, a 24-year old woman, presented with hyperprolactinemia, secondary hypothyroidism, and hypogonadotropic hypogonadism with no evidence of diabetes insipidus. Cerebrospinal fluid (CSF) examination revealed an increased number of lymphocytes and histiocytes. Although β-HCG concentration was normal (<2 mIU/mL) in the CSF, increased β-HCG concentration was detected in the serum. Systemic glucocorticoid treatment led to a decrease in CSF cell count, but no regression of the sellar mass was noted. A diagnostic biopsy was performed and showed an intrasellar germinoma. The patient underwent conventional radiotherapy. Complete resolution of the mass lesion and normalization of β-HCG concentration in the serum were observed three months after radiotherapy. The presence of intrasellar mass lesion in association with pituitary stalk thickening may cause difficulties in the differential diagnosis. Histopathological examination is essential in equivocal cases in order to reach accurate diagnosis and apply the most appropriate therapy.
INTRODUCTION
Intracranial germinomas are malignant neoplasms which most likely arise from primitive germ cells which failed to migrate to the genital crest during embryonic development.1,2 Most of them are located in the suprasellar region and may cause anterior and particularly posterior pituitary hormone deficits.1,3,4 Early establishment of the histological diagnosis is important for optimum treatment planning and a successful outcome. Intracranial germinomas are radiosensitive and potentially curable.1,5 In this communication we describe a patient with an intrasellar germinoma leading to pituitary stalk thickening with lymphohistiocytic reaction, and normal β-HCG concentrations in the cerebrospinal fluid (CSF) but increased β-HCG concentrations in the peripheral blood.
PATIENT DESCRIPTION
A 24-year old woman presented with headache. Physical examination revealed no abnormality. Neurological examination was also normal. Her urine volume and specific gravity were within normal range (urine volume 1800 mL/day, urine specific gravity: 1015). A water deprivation test and plasma AVP measurements were not carried out so that a partial form of diabetes insipidus cannot be totally excluded. Her menstrual history revealed polymenorrhea. Basal hormonal evaluation (by Elecsys-Roche/Hitachi Modular System-Japan) showed hyperprolactinemia, hypogonadotropic hypogonadism, and secondary hypothyroidism (Table 1). Basal serum cortisol concentration indicated that the pituitary-adrenal axis was intact. Magnetic resonance imaging (MRI) revealed an intrasellar mass extending to the suprasellar region, causing diffuse thickening of the pituitary stalk (Figure 1). Because of the thickening of the pituitary stalk, we decided to perform CSF examination to identify the nature of the mass lesion before the patient underwent any invasive procedure. A lumbar puncture was performed and revealed the following (Table 2): Increased protein concentration and increased cell count indicating an inflammatory process. Cytological examination of CSF indicated that the cells were mainly composed of lymphocytes and histiocytes and showed no immunoreactivity against CD1a antigen. Therefore, Langerhans cell histiocytosis could not be verified. Although β-HCG concentration in the CSF was lower than 2 mIU/mL (i.e. within normal limits), increased β-HCG concentration in the peripheral blood obtained simultaneously with the CSF was noted (15 mIU/mL). Serum and CSF α-fetoprotein concentrations were within normal limits. β-HCG determination in the peripheral blood was repeated and the results confirmed the previous finding. β-HCG concentrations were 9.4 mIU/ml and 17.8 mIU/mL on two separate samples. In order to rule out a β-HCG secreting peripheral tumor6 (i.e. bronchogenic, ovarian, hepatic, and renal-cell carcinomas), computerized tomography and magnetic resonance imaging of the chest, abdomen, and pelvis were performed, but no tumor was identified. In addition, gynecologic consultation revealed no abnormality.
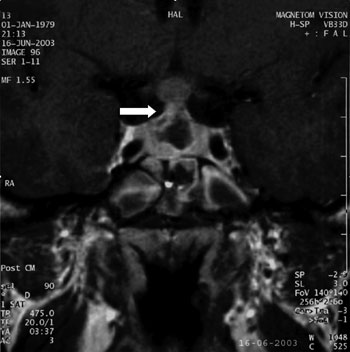
Figure 1.Intra-suprasellar mass lesion leading to stalk thickening (white arrow)
Before the patient underwent transsphenoidal diagnostic procedure, we decided to try a short course of steroid treatment since the CSF findings could be compatible with lymphocytic hypophysitis. While the patient was taking L-thyroxin 50 µg/day for secondary hypothyroidism, methyl prednisolone (equivalent to 1 mg/kg prednisolone) was initiated and MR imaging and CSF examination were repeated 3 weeks later. No regression of intrasellar mass and no resolution of the headache were reported. Repeated CSF examination indicated significant decrease in the cell count (5 cell/mm3) with normal protein concentration. Cytological examination revealed scarce cells within the CSF, while β-HCG concentration was within the normal limit (1.2 mIU/mL), though it was still higher in the peripheral blood (14 mIU/mL). Methyl prednisolone was decreased rapidly and the patient underwent transsphenoidal biopsy. Total tumor removal was not attempted. The histopathological examination confirmed an intrasellar germinoma with strong immunoreactivity for placental alkaline phosphatase. Immunostaining for β-HCG could not be performed because of technical problems. After the operation, conventional radiotherapy with fractional doses for a total of 50 Gy was applied. No other anterior or posterior pituitary hormone deficit developed after the diagnostic procedure. Her serum β-HCG concentration was still higher than normal before the radiotherapy. After radiotherapy, hypocortisolemia developed within a month in addition to previous hormone deficiencies. Glucocorticoids and sex steroids were added to the therapeutic regimen. Repeated sella MRI 3 months after conventional radiotherapy revealed complete resolution of the mass (Figure 2). β-HCG concentration in the peripheral blood had normalized (0.1 mIU/mL). The patient has been well on replacement therapy for hypopituitarism during a follow-up period of 2 years, although mild hyperprolactinemia has persisted. No evidence of recurrence has been observed on serial MRI scans and her serum β-HCG is still <2mIU/mL.
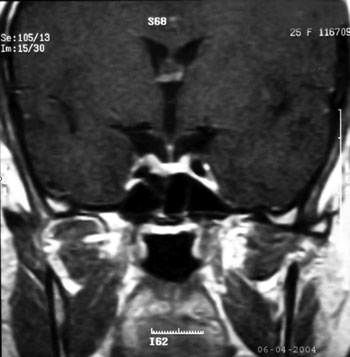
Figure 2.MR imaging of the sella performed 3 months after radiotherapy. No mass lesion was evident.
DISCUSSION
Atypical presentation of intrasellar tumors may create problems in differential diagnosis, as is illustrated by the present case. Diabetes insipidus is considered an early clinical manifestation of germinoma of the sellar region, but it was not a presented sign in our case, although a partial defect in ADH secretion cannot be excluded; urine specific gravity in random specimens was 1015 but a deprivation test was not carried out.
Intrasellar germinomas may cause lymphohistiocytic inflammatory reaction in the CSF and may lead to erroneous diagnosis, such as that of hypophysitis. In addition, systemic anti-inflammatory therapies may have an effect on this type (i.e. tumor associated) of inflammatory reactions and may further delay the definitive diagnosis. Thus, Endo et al and Konno et al reported a 12-year old boy and a 6-year old girl with neurohypophyseal germinomas who presented as granulomatous hypophysitis.8,9 Moreover, a prepubertal girl was reported with biopsy proven hypophysitis and lymphocytic infiltration, which subsequently proved to harbor a germinoma.10 In the latter case, it was suggested that lymphocytic infiltration may represent a first sign of a host reaction to an occult germinoma.
Some intrasellar germinomas cannot interact with the subarachnoidal space, therefore tumor products cannot spill over into the CSF. Communication with the systemic circulation via hypophyseal venous circulation may cause the discrepancy of findings observed in our patient, namely increased β-HCG concentrations in the peripheral blood but normal β-HCG concentrations in the CSF. Although direct proof of this hypothesis could not be established in our case, higher than normal β-HCG in peripheral blood on several occasions and normal CSF β-HCG concentrations on two occasions support this postulation. Normal CSF β-HCG could be attributed to the so-called high dose “Hook” effect. However, we determined β-HCG by Elecsys assay, which is devoid of Hook effect up to 750,000 mIU/mL concentrations of β-HCG.
After radiotherapy, disappearance of the tumor correlated with decreased β-HCG concentrations in the peripheral blood. Seregni et al11 revealed that 12 of their 30 patients with intracranial germ cell tumors had increased serum β-HCG levels, 8 patients had increased β-HCG only in CSF, and 10 patients had no increase in either serum or CSF. It has been reported that increased protein tumor markers in CSF are more sensitive and reliable parameters for the presence of intracranial germ cell tumors.12 Utsuki et al5 evaluated 32 patients with intracranial germinomas for whom HCG/β-HCG levels were measured. In their series, HCG/β-HCG levels were found to be increased in the serum only in 13 patients. In this series, tumors were pineal in 4, suprasellar in 6, and pineal and ventricular in 3 patients. None of the patients had intrasellar localization. Janmohammed et al4 showed that tumor markers were both concomitantly raised in serum and CSF or both undetectable in their patients with suprasellar germ cell tumors. Inamura et al13 indicated that 7 out of 12 patients had increased HCG/β-HCG levels in both serum and CSF. In 5 patients, HCG/β-HCG were negative in the serum but at least one was positive in CSF. In serum-positive cases, localizations were pineal region in 4, basal ganglia in 2, and suprasellar region in one patient. According to these observations, the finding in our patient with intrasellar germinoma of increased β-HCG in the serum and not in CSF is unusual.
Conventional radiotherapy is reported to be associated with mental and pituitary hormonal dysfunction, especially in early childhood.14 In our patient, additional pituitary hormonal deficit (i.e. ACTH deficiency) developed shortly after radiotherapy. A combination of chemotherapy and radiotherapy has been used to reduce the dose of irradiation and radiation induced side effects.15 In addition, stereotactic radiotherapy is applied to minimize the late radiation morbidity.16 Long-term results of these treatment modalities are required to fully determine their efficacy and side effects.
Pituitary stalk thickening on MRI suggests infectious, inflammatory or granulomatous lesions. Lymphocytic infundibuloneurohypophysitis –an autoimmune inflammatory disorder—causes central diabetes insipidus and pituitary stalk thickening which may later disappear.14 Langerhans cell histiocytosis, tuberculosis, sarcoidosis, Wegener granulomatosis, and granulomatous hypophysitis may also cause pituitary stalk thickening. Various neoplastic conditions, such as germinomas, metastatic lesions, leukemic infiltration, lymphoma, teratoma, craniopharyngioma, and hypothalamic glioma, may also cause stalk thickening.17,18 CSF examination may be helpful for the definitive diagnosis. It has been suggested that because germinomas usually have a suprasellar localization, diabetes insipidus is present at the onset of the disease in most patients.3,4,7 However, in our patient with involvement of pituitary stalk and hyperprolactinemia, no clinical findings of overt diabetes insipidus developed. Although careful analysis of clinical, laboratory, and imaging findings may facilitate the diagnosis in most of the cases with intrasellar masses and pituitary stalk thickening, transsphenoidal biopsy should be performed in equivocal cases. Correct diagnosis allowed us to carry out the most appropriate treatment.
REFERENCES
1. Packer RJ, Cohen BH, Coney K, 2005 Intracranial germ cell tumors. Oncologist 5: 312-320.
2. Horowitz MB, Hall WA, 1991 Central nervous system germinomas: a review. Arch Neurol 48: 652-657.
3. Mootha SL, Barkovich AJ, Grumbach MM, et al, 1997 Idiopathic hypothalamic diabetes insipidus, pituitary stalk thickening and the occult intracranial germinoma in children and adolescents. J Clin Endocrinol Metab 82: 1362-1367.
4. Janmohamed S, Grossman AB, Metkalfe K, et al, 2002 Suprasellar germ cell tumours: specific problems and the evolution of optimal management with a combined chemoradiotherapy regimen. Clin Endocrinol 57: 487-500.
5. Utsuki S, Oka H, Tanaka S, Tanizaki Y, Fujii K, 2002 Long-term outcome of intracranial germinoma with hCG elevation in cerebrospinal fluid but not in serum. Acta Neurochir 144: 1151-1155.
6. HirsbergB, Conn M, Uwaifo GI, Blauer KL, Clark BD, Nieman LK, 2003 Ectopic luteinizing hormone secretion and anovulation. New Eng J Med 348: 312-317.
7. Buchfelder M, Fahlbusch R, Walther M, Mann K, 1989 Endocrine disturbances in suprasellar germinomas. Acta Endocrinol 120: 337-342.
8. Endo T, Kumabe T, Ikeda H, Shirane R, Yoshimoto T, 2002 Neurohypophyseal germinoma histologically misidentified as granulomatous hypophysitis. Acta Neurochir 144: 1233-1237.
9. Konno S, Oka H, Utsuki S, Kondou K, Tanaka S, Fujii K, Yagishita S, 2002 Germinoma with a granulomatous reaction. Problems of differential diagnosis. Clin Neuropathol 21: 248-251.
10. Bettendorf M, Fehn M, Grulich-Henn J, et al, 1999 Lymphocytic hypophysitis with central diabetes insipidus and consequent panhypopituitarism preceding a multifocal intracranial germinoma in a prepubertal girl. Eur J Pediatr 158: 288-292.
11. Seregni E, Massimino M, Nerini Molteni S, et al, 2002 Serum and cerebrospinal fluid human chorionic gonadotropin (hCG) and alpha-fetoprotein (AFP) in intracranial germ cell tumors. Int J Biol Markers 17: 112-118.
12. Allen JC, Nisselbaum J, Epstein F, Rosen G, Schwarts MK, 1979 Alpha-fetoprotein and human chorionic gonadotropin determination in cerebrospinal fluid: an aid to the diagnosis and management of intracranial germ cell tumors. J Neurosurg 51: 368-374.
13. Inamura T, Nishio S, Ikezaki K, Fukui M, 1999. Human chorionic gonadotrophin in CSF not serum predicts outcome in germinoma. J Neurol Neurosurg Psychiatry 66: 654-657.
14. Oka H, Kawano N, Tanaka T, et al, 1998 Long-term functional outcome of suprasellar germinomas: usefulness and limitations of radiotherapy. J Neurooncol 40: 185-190.
15. Matsutani M, Sano K, Takakura K, Fujimaki T, Nakamura O, 1998 Combined treatment with chemotherapy and radiation therapy for intracranial germ cell tumors. Child’s Nerv Syst 14: 59-62.
16. Zissiadis Y, Dutton S, Kieran M, et al, 2001 Stereotactic radiotherapy for pediatric intracranial germ cell tumors. Int J Radiat Oncol Biol Phys 51: 108-112.
17. Kanagaki M, Miki Y, Takahashi JA, et al, 2004 MRI and CT findings of neurohypophyseal germinoma. Eur J Radiol 49: 204-211.
18. Shin JH, Lee HK, Choi CG, Suh DC, Hong SK, Na DG, 2001 MR imaging of central diabetes insipidus. A pictorial essay. Korean J Radiol 2: 222-230.
Address correspondence and requests for reprints to:
Nese Ozbey, Tozkoparan Mah, Kopuz Sokak, Cevahir Koru Sitesi,
D Blok, Daire: 10, Merter, 34010, Istanbul/Turkey,
Tel.: + 90 212 482 80 40, + 90 542 564 14 54 (GSM),
Fax: + 90 212 635 85 22
Received 31-10-05, Revised 27-11-05, Accepted 06-12-05