Abstract
Objective: Measurement of cortisol levels in saliva is a marker of free hormone. How salivary cortisol rhythm is affected by age, gender, the metabolic syndrome and estrogen-progestin therapy was evaluated in a community sample of adults.
Subjects and Methods: One hundred twenty volunteers recruited from the Hospital staff and family members of the Endocrinology Unit were instructed to collect 7 salivary samples: the first on awakening (F0) and 6 more (F1.5, F5, F6, F10, F11.5 and F14) over the next 14 hours. Each volunteer also underwent a complete physical evaluation and a comprehensive medical history was taken. Salivary cortisol was measured using a radioimmunometric assay. Daily cortisol secretion was evaluated computing the Area Under the Curve (AUCF0→F14); the F14/F0 ratio was calculated as a marker of cortisol rhythm.
Results: Median F14 levels were higher in the subjects in the third tertile of age than in those falling in the second or in the first age tertile (respectively, 2.09 vs 1.33 vs 1.25 ng/mL, p=0.023 and p=0.006), in the hypertensive volunteers (2.44 vs 1.44 ng/mL, p=0.030) and in those with the metabolic syndrome (2.95 vs 1.4 ng/mL, p=0.002), with an elevated median F14/F0 ratio (0.48 vs 0.19, p=0.006). According to the Kruskal-Wallis analysis of variance, the most important factor affecting F14 value was age (p=0.001). AUCF0→F14 was not influenced by gender, age, metabolic syndrome or estrogen-progestin therapy.
Conclusions: While it did not affect the daily cortisol rate, late-night salivary cortisol levels were found to be increased in the subjects in the higher age tertile and in those with the metabolic syndrome.
INTRODUCTION
Regardless of the rate of saliva production, the concentration of cortisol in saliva parallels free biologically active plasma cortisol levels.1 Corticosteroids in saliva were first measured by an Italian study in the 1960s,2 but it was only in the late 1980s that reports began to appear in the literature outlining the clinical utility of salivary cortisol measurements.3 Salivary cortisol sampling is now increasingly used to test for hypothalamic-pituitary-adrenal (HPA) axis disorders3 and to titrate substitutive therapy in adrenal insufficiency.4
The noninvasive and readily repeatable nature of the assay makes salivary cortisol an ideal tool for investigating circadian rhythm. According to a large population-based study, morning salivary cortisol is negatively associated with Body Mass Index (BMI) and waist circumference,5 with males showing a higher secretion rate than females.1 Salivary sampling is particularly useful when free cortisol measurements are needed, i.e. in medical conditions characterized by increased cortisol-binding globulin levels, such as during pregnancy or when estrogen-progestin contraceptives are being taken.6,7
The aim of this study was to examine, in a community sample of adults, whether, and if so how, age, gender, metabolic syndrome and estrogen-progestin therapy affect salivary cortisol rhythm.
MATERIAL AND METHODS
Adult volunteers
One hundred twenty adult Hospital employees and the family members of the Endocrinology Unit of the University of Padova Medical Centre were recruited (51 males and 69 females); we excluded those who had performed night shifts one month before salivary collection in order to avoid cortisol rhythm impairment. None of these showed any clinical signs of hypercortisolism, were taking drugs that could interfere with the HPA axis or had a clinical history of adrenal incidentaloma or liver/renal failure.
Wearing light clothing and no shoes, participants were weighed and measured using a balanced beam scale and a vertical ruler. Weight was recorded to the nearest 0.5 kg and height to the nearest 0.5 cm. BMI was calculated (weight divided by height squared, kg/m2) and on the basis of these measurements the participants were divided into 4 BMI classes: ≤20 kg/m2 (21 subjects, considered underweight), 20.1-25 kg/m2 (63 subjects, considered normal), 25.1-30 kg/m2 (30 subjects, considered overweight) and >30 kg/m2 (6 subjects, considered obese). In accordance with the WHO guidelines, waist circumference was measured at the end of natural breaths at the midpoint between the top of the iliac crest and the lower margin of the last palpable rib (<94 cm was considered normal in males and <80 cm in females). Hip circumference was measured around the widest part of the buttocks.8 We also calculated the Waist-to-Hip Ratio (WHR is waist circumference divided by hip circumference) which was considered high if it was ≥0.9 in males and ≥0.85 in females.8 We considered subjects hypertensive if home systolic blood pressure levels were ≥130 mmHg or diastolic ≥85 mmHg (or if they were receiving antihypertensive treatment). The subjects’ lipid profiles were also assessed and they were considered dyslipidemic if plasma triglycerides were ≥1.7 mmol/L or HDL <1 mmol/L in males and <1.3 mmol/L in females (or being treated for dyslipidemia). The participants were considered diabetic if fasting plasma glucose was ≥5.5 mmol/L (or they were taking antidiabetic drugs). The metabolic syndrome, defined as a cluster of risk factors (increased waist circumference, elevated triglycerides or reduced HDL levels, hypertension and diabetic mellitus), was also assessed. We defined the participants as having the metabolic syndrome if they had at least 3 out of 5 of the risk factors listed.9
The study was performed in accordance with the principles outlined in the Declaration of Helsinki: the local Ethics Committee approved the study protocol and all the subjects gave informed consent.
Saliva Collection
The volunteers were asked to collect 7 saliva samples during an ordinary weekday: the first sample was to be collected without delay upon awakening (labeled F0, hours of awakening from 6.30 to 7.30am), an hour and 30 minutes later (F1.5), then 5 hours (F5), 6 hours and 30 minutes (F6.5), 10 hours (F10), 11 and 30 minutes after (F11.5) and finally 14 hours after the first collection (F14, which corresponded to late-night salivary cortisol). The subjects were instructed not to eat, drink or to brush their teeth (to avoid any source of food or blood contamination) for at least 30 minutes before taking each saliva sample. Subjects had lunch between F5 and F6.5, and dinner between F11.5 and F14. They were also asked to avoid smoking or eating liquorice on the day saliva was being collected.
Saliva was collected, stored and analyzed as described elsewhere.4 Briefly, subjects were asked to collect samples using the Salivette commercial device (Salivette®, Sarstedt, Numbrecht, Germany). Absorbent synthetic swab rolls were to be soaked in the saliva for 2-3 min and then placed in syringes and kept in the participant’s refrigerator. Participants were asked to send the samples to our center the day after sampling and upon receipt they were kept at +4°C, centrifuged and stored at -20°C until assayed with a commercially available RIA kit (Radim, Rome, Italy), which has an assay sensitivity of 0.5 ng/mL and an intra-assay and inter-assay variation of 3% and 9%. Regarding specificity, this cortisol assay does not show significant cross-reaction with cortisone, desoxycorticosterone, corticosterone, estradiol, testosterone, 17 hydroxyprogesterone; the coefficient of variation in the high and in the low range were, respectively, 7% and 4% (local data).
Statistical Analysis
Proportions and rates were analyzed for categorical data and continuous data are expressed as median and interquartile range (IQR). To assess the total endogenous daily cortisol secretion, the Area Under the Curve with respect to ground (AUCF0→F14) of F0 and F14 was computed according to the trapezoidal method;10 moreover, the F14/F0 ratio was also calculated as an estimation of cortisol rhythm. We also calculated percentiles, defined as the value that divides the distribution in such a way that a specific percentage of data is ≤ the defined percentile calculated with the NIST (National Institute of Standards and Technology) formula. The groups were compared using the chi-square test for categorical variables (or Fisher’s exact test when cell count was <5) and using the Mann-Whitney test for quantitative data (age, BMI, salivary sample Fn, F14/F0 ratio, AUCF0→F14); we analyzed the relationship between two variables via linear regression. We calculated the contribution of age, sex, BMI, WHR, hypertension and metabolic syndrome to the F14 salivary cortisol levels with the Kruskal-Wallis one-way analysis of variance by ranks test. The level of significance was set at p <0.05 for all tests. Database management and statistical analysis were performed using the SPSS 17 software package (SPSS Inc, Chicago, IL).
RESULTS
Gender and age difference
There were no gender differences in salivary cortisol levels at the different time-points either in the F14/F0 ratio or in the AUCF0→F14.
The participants were divided into 3 age tertiles: first tertile (38 subjects, age <26 years), second tertile (43 subjects, age 27-45 years) and third tertile (39 subjects, age >46 years). There were differences in the night salivary cortisol levels in the different age classes: median F14 levels were higher in the participants in the third tertile of age than those in the second [2.09 (IQR 1.32-3) vs 1.33 ng/mL (IQR 0.65-2.24), p=0.023] and in the first tertile of age distribution [2.09 (IQR 1.32-3) vs 1.25 (IQR 0.5-2.1) ng/mL, p=0.006)], as depicted in Figure 1. Linear regression analyses showed an age-related increase in late-night salivary cortisol (F14) (y = 0.0324x+0.5482, R2 0.17, p=0.009; dependent variable F14 and independent variable age in years). The median F14/F0 ratio was higher in the subjects in the third tertile of age than those in the first [0.26 (IQR 0.15-0.46) vs 0.18 (IQR 0.10-0.25), p=0.035].
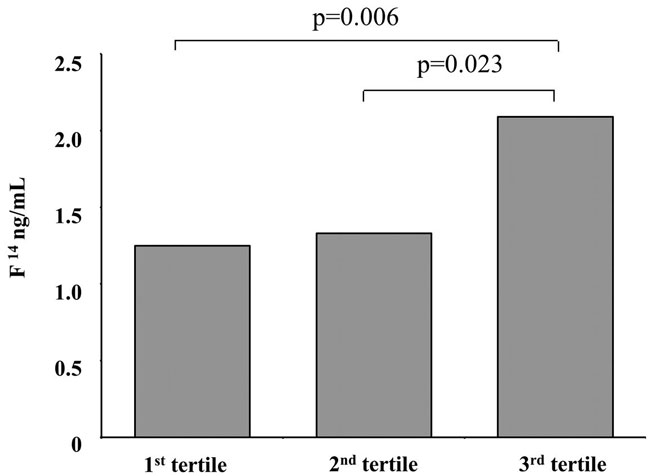
Figure 1. Median levels of late-night salivary cortisol (F14) in the participants grouped into age brackets.
We excluded endogenous Cushing’s syndrome in the participants whose late-night salivary cortisol levels were higher than our cut-off (5.24 ng/ml),11 because their serum cortisol levels after a 1-mg overnight dexamethasone suppression test were <50 nmol/L (the lower cut-off, in order to avoid false negative results) and the mean of two 24-hour urinary free cortisol collections were <170 nmol/24h (our local cut-off for endogenous hypercortisolism.12
The percentiles of salivary cortisol at each of the 7 time-points and the AUCF0→F14 are outlined in Table 1. We did not observe an increase in cortisol levels after meals.
Role of the metabolic syndrome
There were no differences in salivary cortisol levels at the different time-points either in the F14/F0 ratio or in the AUCF0→F14 in the underweight, normal, overweight and obese subjects.
With regard to gender-based cut-offs (<94 males and <80 cm in females), waist circumference was normal in 65 subjects (32 males and 33 females) and high in 55 volunteers (19 males and 36 females): there were no differences in salivary cortisol levels at the different time-points either in the F14/F0 ratio or in the AUCF0→F14 in the subjects with normal or high gender-adjusted waist circumference. When different gender cut-offs for WHR were used (≥0.9 in males or ≥0.85 in females),8 there were no differences in salivary cortisol levels at the different time-points either in the F14/F0 ratio or in the AUCF0→F14 in the subjects with normal (total 48 subjects, 15 males and 33 females) or high WHR (total 72 subjects, 36 males and 36 females) groups.
Both median F11.5 [2.08 (IQR 1.6-3) vs 1.4 (IQR 0.5-2.26) ng/mL, p=0.022] and F14 [2.44 (IQR 1.39-3) vs 1.44 (IQR 0.56-2.22) ng/mL, p=0.030] were higher in the 16 hypertensive patients (all receiving medical therapy); the salivary cortisol levels at the other time-points, the F14/F0 ratio and the AUCF0→F14 were found to be similar in the normal and hypertensive subjects.
Late-night salivary cortisol and the F14/F0 ratio were higher in the patients with the metabolic syndrome with respect to those without [median F14 levels were 2.95 (IQR 2.31-3.54) vs 1.4 (IQR 0.56-2.22) ng/mL, p=0.002; median F14/F0 ratio was 0.48 (IQR 0.28-0.57) vs 0.19 (IQR 0.12-0.33), p=0.006]: those patients with ≥3 risk factors of the metabolic syndrome (n=8) had higher median F14 levels than those with 1-2 risk factors (n=52) or no risk factors [respectively, 2.95 ng/mL (IQR 2.31-3.00), 1.52 ng/mL (IQR 0.60-2.22) and 1.32 (IQR 0.50-1.22) ng/mL, p=0.006 and p=0.002]. F14 levels were, instead, similar in the subjects with no or 1-2 risk factors (Figure 2). Similarly, the median F14/F0 ratio was higher in the patients with more than 3 risk factors than in those 1-2 or 0 risk factors (respectively, 0.48 (IQR 0.25-0.55), 0.20 (IQR 0.13-0.34) and 0.18 (IQR 0.10-0.32); p=0.013 in the 3 risk factors vs 1-2 risk factors and p=0.005 in the 3 risk factors vs no risk factors), while the F14/F0 ratio was similar in the no risk factor and 1-2 risk factors groups.
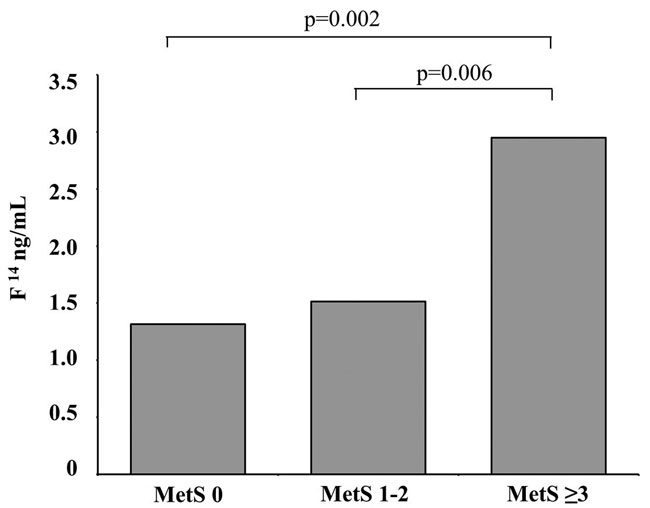
Figure 2. Median levels of late-night salivary cortisol (F14) in the participants grouped in accordance with metabolic syndrome features.
Estrogen-progestintherapy
We also compared the results of the 21 women who were taking estrogen-progestin contraceptives (17 oral and 4 transdermal) with a group of 31 women aged ≤45 years, reportedly having regular menses, and collected salivary samples during their follicular phase. There were no differences in salivary cortisol levels at the various time-points either in the F14/F0 ratio or in the AUCF0→F14 in these two groups.
Analysis of variance
On the basis of the Kruskal-Wallis one-way analysis of variance of all the factors described (age, sex, BMI, WHR, metabolic syndrome, hypertension), only age, hypertension and metabolic syndrome had a significant effect on F14 levels, with the most powerful p value linked to age (X2=19.101; p=0.004) rather than to hypertension (X2=17.537; p=0.007) and metabolic syndrome (X2=16.287; p=0.012); the latter factors were not significant after adjusting for the other variables (age). The participants with metabolic syndrome were, indeed, older than those without [median 71 (IQR 54-81) vs 30 (IQR 26-48) years, p <0.001] and those with ≥3 risk factors [median 71 (IQR 54-81) years] were older than those with 1-2 [median 45 years (IQR 31-55), p=0.002] and those with none [median 26 years (IQR 24-30), p <0.001]. Also, median age of hypertensive volunteers [65 years (IQR 48-80)] was higher than normotensive subjects [30 years (IQR 26-45), p<0.001].
DISCUSSION
The clinical utility of measuring salivary cortisol and of calculating its daily production rate has recently been confirmed by several studies.3-5,11,13We asked unselected subjects living in the community to collect 7 saliva samples on an ordinary weekday in order to define the percentiles of physiological cortisol concentrations at any time-point of the day and the AUCF0→F14, a mathematical method used to incorporate multiple time-points with an acceptable degree of approximation.10 Our results confirmed the percentiles of salivary cortisol described elsewhere.4As there is as yet no consensus about how to best manage adrenal insufficiency,13,14 these data could be useful to titrate glucocorticoid therapy in these patients.
Our findings confirmed that salivary cortisol AUCF0F14 is not influenced by gender, age, metabolic syndrome or the use of estrogen-progestin contraceptives in adult subjects. This may seem surprising, as some studies have reported that cortisol levels increase with age15 and that older age is associated with higher daily cortisol secretion.16 In accordance with the findings outlined by Pecori Giraldi et al,17 our study confirmed an age-related increase only for evening and late-night salivary cortisol, leading, as a consequence, to a higher night/morning cortisol ratio (F14/F0), a simple marker of impaired circadian rhythm which shows a blunt decline in elderly volunteers. We did not, however, find any age-related modification in AUCF0→F14, probably because evening levels were not sufficiently increased to alter the global daily secretion or because our cohort was not as large as those previously described. In this line of evidence, we recently reported that also urinary free cortisol levels, which could be considered another integrated marker of daily cortisol secretion rate, are not affected by aging.18
In contrast to another study,1 we did not find any gender differences in salivary cortisol values, probably because our volunteers included individuals from 20 to 88 years old, while Lederbogen et al. studied only 50-69 year-old subjects.1 As likewise reported by Manetti et al,7 we found no differences in late-night salivary cortisol levels, in daytime cortisol rhythm, as indicated by the F14/F0 ratio, or in cortisol production rate, as indicated by AUCF0→F14, between the women of reproductive age with regular menses who collected saliva during their follicular phase and those who were on estrogen-progestin therapy. In view of the fact that salivary cortisol reflects the free fraction of the hormone, saliva testing could be a particularly useful tool in the clinical setting in which cortisol-binding globulin is elevated.
We found higher late-night salivary cortisol levels in the hypertensive subjects and in those with metabolic syndrome; the latter were also characterized by a slow cortisol decline, reflected in a higher F14/F0 ratio. In our volunteers, 4 out of 120 subjects (3%) had late-night salivary cortisol levels that were higher than our local upper cut-off:11 they were all in the third tertile of age distribution, one was overweight, 2 were obese and 3 were hypertensive. We used the 1-mg overnight dexamethasone suppression test and a 24-hour free urinary cortisol collection to exclude Cushing’s syndrome in all 4 cases. A recent study reported that salivary cortisol was negatively associated with BMI and waist circumference and that cortisol secretion was lower in the overweight subjects during the first part of the day.5 We observed higher late-night salivary cortisol levels only in the subjects with metabolic syndrome (with at least 3 risk factors). This observation is of clinical interest because it is well known that a delayed cortisol rhythm with relatively slight cortisol excess in the evening, obtained in metyrapone-suppressed healthy volunteers treated with a physiological hydrocortisone dose, is linked to the worsening of glucose metabolism (increase in glucose levels and insulin concentrations).19 The interplay between the metabolic syndrome and subtle cortisol secretion alterations, characterized as subclinical Cushing’s syndrome, has been reported in patients with adrenal incidentaloma: a slight cortisol excess is associated with an increased prevalence of the metabolic syndrome and possibly cardiovascular impairment that improves after adrenalectomy.20,21
According to our findings, only age has a significant effect on late-night salivary cortisol levels. As the subjects with hypertension or metabolic syndrome were older than those without, this reflected an increasing incidence of hypertension and metabolic syndrome with age.22 Forming a differential diagnosis between Cushing’s syndrome and metabolic syndrome can be a challenge for the endocrinologist due to overlapping clinical features in the two conditions. Impaired cortisol rhythm in Cushing’s syndrome,23 which is easily studied using night salivary cortisol determinations,3,24 might at times be misleading in older patients with the metabolic syndrome. As there is the risk of false positive test results in these cases, other screening tests should be utilized to identify the syndrome. This situation is frequently observed when biochemical workup is being carried out where there is suspicion of adrenal incidentaloma, whose incidence, just like metabolic syndrome, increases with age (with a peak around the fifth and seventh decade).25 In contrast to the other larger population study,1,5 the present one has the advantage that all participants exhibiting elevated late-night salivary cortisol levels performed a dexamethasone suppression test to exclude endogenous hypercortisolism, which is difficult to perform in population-based settings; moreover, we used the lower cut-off (serum cortisol levels were <50 nmol/L) to avoid false negative testing. Furthermore, there is an extensive body of literature on salivary cortisol measuring, but most of the studies focus on morning cortisol/awakening response: our focus on cortisol rhythm provides new data to a less extensively studied field.
Our study has some limitations. First of all, the analytical specificity and sensitivity of assay methods may vary: many of the commercial assays available to measure salivary cortisol can produce false values due to cross-reactivity with metabolites such as cortisone. Thus, the general trends of values presented are probably not transferable to other assay systems that are widely used in clinical practice. We have, nevertheless, already described the effectiveness of salivary cortisol in detecting Cushing’s syndrome in a large cohort of patients and control subjects. Though adult volunteers were recruited, we did not study the population prospectively. In this sample, Hospital employees and their family members were recruited, and this may be a disadvantage due to the possible similarity in cortisol diurnal rhythm among relatives.
To conclude, study findings showed that the salivary cortisol AUC is not influenced by age, gender, metabolic syndrome or use of estrogen-progestin therapy. In contrast, only late-night salivary cortisol levels were found to increase with age and in patients with the metabolic syndrome. Endocrinologists should bear in mind that elderly subjects with adrenal incidentaloma or suspicion of Cushing’s syndrome can produce false positive salivary cortisol test results.
DISCLOSURE
The authors have no conflicts of interest to declare, nor any financial interests to disclose.
REFERENCES
1. Lederbogen F, Kühner C, Kirschbaum C, et al, 2010 Salivary cortisol in a middle-aged community sample: results from 990 men and women of the KORA-F3 Augsburg study. Eur J Endocrinol 163: 443-451.
2. Casella E, 1968 Variations in the salivary concentrations of cortisol and cortisone in normal women and during pregnancy. Dental Cadmos 36: 1623-1639.
3. Raff H, 2012 Cushing’s syndrome: diagnosis and surveillance using salivary cortisol. Pituitary 15: 64-70.
4. Ceccato F, Albiger N, Reimondo G, et al, 2012 Assessment of glucocorticoid therapy with salivary cortisol in secondary adrenal insufficiency. Eur J Endocrinol 167: 769-776.
5. Champaneri S, Xu X, Carnethon MR, et al, 2013 Diurnal salivary cortisol is associated with body mass index and waist circumference: the Multiethnic Study of Atherosclerosis. Obesity 21: E56-E63.
6. Goodman MP, 2012 Are all estrogens created equal? A review of oral vs. transdermal therapy. J Womens (Larchmt) Health 21: 161-169.
7. Manetti L, Rossi G, Grasso L, et al, 2013 Usefulness of salivary cortisol in the diagnosis of hypercortisolism: comparison with serum and urinary cortisol. Eur J Endocrinol 168: 315-321.
8. Waist Circumference and Waist–Hip Ratio Report of a WHO Expert Consultation 2008, available at http://whqlibdoc.who.int/publications/2011/9789241501491_eng.pdf
9. Alberti KG, Eckel RH, Grundy SM, et al, 2009 Harmonizing the metabolic syndrome: a joint interim statement of the International Diabetes Federation Task Force on Epidemiology and Prevention; National Heart, Lung, and Blood Institute; American Heart Association; World Heart Federation; International Atherosclerosis Society; and International Association for the Study of Obesity. Circulation 120: 1640-1645.
10. Pruessner JC, Kirschbaum C, Meinlschmid G, et al, 2003 Two formulas for computation of the area under the curve represent measures of total hormone concentration versus time-dependent change. Psychoneuroendocrinology 28: 916-931.
11. Ceccato F, Barbot M, Zilio M, et al, 2013 Performance of salivary cortisol in the diagnosis of Cushing’s syndrome, adrenal incidentaloma, and adrenal insufficiency. Eur J Endocrinol 169: 31-36.
12. Antonelli G, Artusi C, Marinova M, et al, 2014 Cortisol and cortisone ratio in urine: LC-MS/MS method validation and preliminary clinical application. Clin Chem Lab Med 52: 213-220.
13. Grossman AB, 2010 The Diagnosis and Management of Central Hypoadrenalism. J Clin Endocrinol Metab 95: 4855-4863.
14. Debono M, Price JN, Ross RJ, 2009 Novel strategies for hydrocortisone replacement. Best Pract Res Clin Endocrinol Metab 23: 221-232.
15. Peeters GM, van Schoor NM, van Rossum EF, et al, 2008 The relationship between cortisol, muscle mass and muscle strength in older persons and the role of genetic variations in the glucocorticoid receptor. Clin Endocrinol 69: 673-682.
16. Nater UM, Hoppmann CA, Scott SB, 2013 Diurnal profiles of salivary cortisol and alpha-amylase change across the adult lifespan: Evidence from repeated daily life assessments. Psychoneuroendocrinology 38: 3167-3171.
17. Pecori Giraldi F, Ambrogi AG, De Martin M et al, 2007 Specificity of first-line tests for the diagnosis of Cushing’s syndrome: assessment in a large series. J Clin Endocrinol Metab 92: 4123-4129.
18. Ceccato F, Antonelli G, Barbot M, et al, 2014 The diagnostic performance of urinary free cortisol is better than the cortisol/cortisone ratio in detecting de novo Cushing’s syndrome: the use of a LC-MS/MS method in routine clinical practice. Eur J Endocrinol 171: 1-7.
19. Plat L, Leproult R, L’Hermite-Baleriaux M, et al, 1999 Metabolic effects of short-term elevations of plasma cortisol are more pronounced in the evening than in the morning. J Clin Endocrinol Metab 84: 3082-3092.
20. Iacobone M, Citton M, Viel G, et al, 2012 Adrenalectomy may improve cardiovascular and metabolic impairment and ameliorate quality of life in patients with adrenal incidentalomas and subclinical Cushing’s syndrome. Surgery 152: 991-997.
21. Chiodini I, Morelli V, Salcuni AS, et al, 2010 Beneficial metabolic effects of prompt surgical treatment in patients with an adrenal incidentaloma causing biochemical hypercortisolism. J Clin Endocrinol Metab 95: 2736-2745.
22. Eckel RH, Grundy SM, Zimmet PZ, 2005 The metabolic syndrome. Lancet 365: 1415-1428.
23. Nieman LK, Biller BM, Findling JW, et al, 2008 The diagnosis of Cushing’s syndrome: an Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab 93: 1526-1540.
24. Carroll T, Raff H, Findling JW, 2009 Late-night salivary cortisol for the diagnosis of Cushing syndrome: a meta-analysis. Endocr Pract 15: 335-342.
25. Terzolo M, Stigliano A, Chiodini I, et al, 2011 AME position statement on adrenal incidentaloma. Eur J Endocrinol 164: 851-870.
Address for correspondence:
Filippo Ceccato, MD, Endocrinology Unit, Department of Medicine DIMED; Via Ospedale Civile, 105 35128 Padova, Italy; Tel.: +39 049 8213000; Fax: +39 049 657391; E-mail: ceccato.filippo@gmail.com
Received 08-08-2014, Accepted 02-03-2015